reSee.it - Tweets Saved By @CanningPharm

@CanningPharm - MariaGutschi✝️📿💊👵🦠
1/ What do we have here? From @sco0psmcgoo ATIP we have the following from Dr Tong Wu, written Sept 5, 2023. Here she is reminding Pfizer regarding their obligation to report Out Of Specifications for stability monitoring. https://t.co/R4rVqe9FNQ
@CanningPharm - MariaGutschi✝️📿💊👵🦠
2/ OOS is pretty serious. This meets it does not meet specification on one or more critical quality attributes. For final product testing this means stuff like quantity of RNA potency tests LNP size and range of sizes etc Since this is for stability monitoring I would guess LNP size or mRNA quantity
@CanningPharm - MariaGutschi✝️📿💊👵🦠
3/ OHHH look at this https://www.drug-inspections.canada.ca/gmp/fullReportCard-en.html?lang=en&insNumber=81383 Health Canada did an inspection of the Pfizer facility in Kirkland Quebec on August 7, 2023. Remember on August 4th, 2023 Tong Wu and team sent out their first clarifax to Pfizer, and the response back from Pfizer on August 5th. The last time they did an in-person inspection was August 26, 2019 since before the pandemic. hmmmmm....
@CanningPharm - MariaGutschi✝️📿💊👵🦠
4/ Let's look at the inspection report. It says Pfizer was compliant BUT the report is not what I would call ahem good. 1. storage and transportation issues? Not keeping stuff at the right temperature? Logging shipments appropriately? Keeping the cold chain? 2. evaluation was inadequate for cases where there was a deviation from the norm and/or where products were on the borderline of the acceptable range. Ahhh, you mean they just wrote off OOS results as OK? Seriously? 3. this is my favourite though The assessment, recording, follow-up, and/or investigation of complaints and/or other information about potentially defective products was inadequate. Hahahah. So that means complaints from pharmacists and others about say visible particles, labelling issues, problems with the stoppers, expired products, cold chain not kept was ignored? Good to know.
@CanningPharm - MariaGutschi✝️📿💊👵🦠
5/ So after 3.5 years of manufacturing, and specifications which were wide enough to drive a truck through at the beginning are STILL not being met in August 2023? Maybe SOME specifications have been tightened since the first EMA leak/hack but still. I have to ask why is this still happening? @NChartierET @DJSpeicher @AdhesionsOrg @Kevin_McKernan @JesslovesMJK

@CanningPharm - CanningPharmD✝️📿💊👵🦠
1/The pharmacokinetics of LNPs are exceedingly complex but this paper is the best so far in trying to apply traditional physiologic-based pharmacokinetic modelling with some empirical validation. Let's see if I can explain it to the best of my ability (note paper at end of thread)
@CanningPharm - CanningPharmD✝️📿💊👵🦠
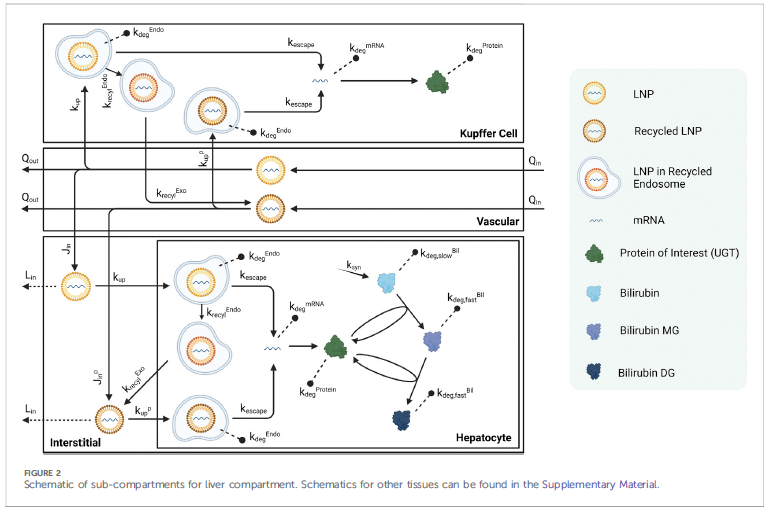
2/That figure is a schematic of a liver Kupffer cell, blood or vascular flow, a hepatocyte and interstitial fluid. through the liver 2. LNPs passively infiltrate the interstitial space and can either be taken up by the Kupffer cells or hepatocyte (transfection) 3. or flow back into lymph nodes and then back again into the vasculature and are recycled 4. once the LNP transfects either the Kupffer or hepatocytes they can undergo one of 4 processes
@CanningPharm - CanningPharmD✝️📿💊👵🦠
3/ a. degradation through the endosomes and then lysosomes (can take a long time) and are stuck there b. mRNA escape into the cytoplasm (the desired effect), then production and degradation of the expressed protein c. transition into recycled endosomes and exocytosed d. degradation of escaped mRNA in the cell These exocytosed LNPs likely do not have the same surface properties of the original LNPs but can still travel and transfect other cells, and are the cause of "shedding" imho. Also, the recycled but plain LNPs which travel through the vascular and lymph circulation produce a second peak in LNP kinetics sometimes seen experimentally
@CanningPharm - CanningPharmD✝️📿💊👵🦠
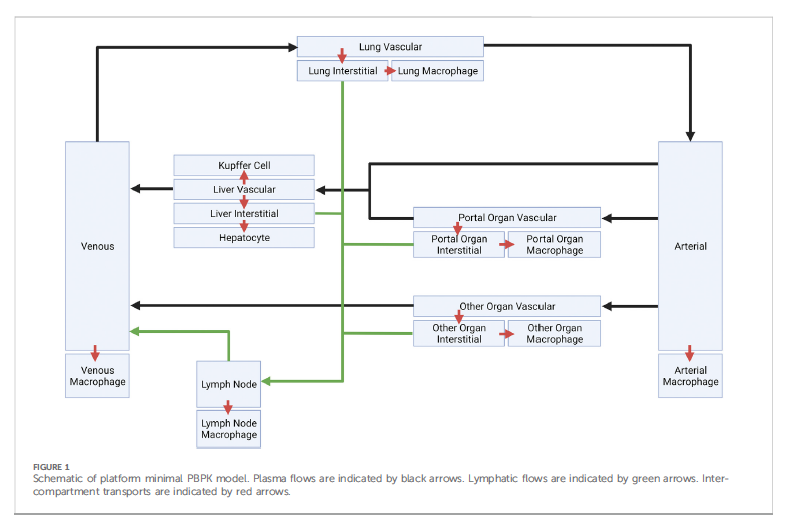
4/This is the overall physiologic-based pharmacokinetic model scaled down with only 7 compartments (my head explodes after 3!!). In this case the mRNA/LNP is given intravenously and you can see the compartments considered "major". Then within each of these compartments (or grey grouped-boxes), you would go through the exercises above and add mathematical formulas describing the flow, degradation and other rates.
@CanningPharm - CanningPharmD✝️📿💊👵🦠
5/ So even with a scaled down model with 7 compartments, each compartment with about 11-12 parameters plus, and each parameter with a variable rate between people or even between doses, you can see the extreme variability which can occur, most of which one cannot measure in vivo. So easily 1-100 fold difference in say protein production. Or more.
@CanningPharm - CanningPharmD✝️📿💊👵🦠
6/ This data is from a study by Moderna for their enzyme replacement therapy for Crigler-Najjar (CN) syndrome which they have been working on for some time. The model is most positively sensitive to the endosomal and mRNA degradation rates (100% increase for both), followed by the mRNA translation and escape rates (around 50% increase). It is moderately sensitive to the cellular uptake rate, but not sensitive to the transport rate from vascular into interstitial space. So biodistribution per se doesn't really affect protein production that much or even transfection, it is ESCAPE from the endosome and a slowly degrading modRNA. HMMMMMM
@CanningPharm - CanningPharmD✝️📿💊👵🦠
7/ Here's the paper. What does everyone think? Or do you hate nanoparticles more than ever, lolol? https://www.frontiersin.org/articles/10.3389/fnano.2024.1330406/full#B16


@CanningPharm - CanningPharmD✝️📿💊👵🦠
What are the lies about the vax that bother me. 1. mRNA degrades rapidly 2. The spike protein degrades rapidly 3. The mRNA is just like your mRNA in your body 4. It makes the same spike protein as the virus 5. It stays in the muscle 6. No short cuts were taken in the regulatory review 7. It’s a vaccine like the flu vaccine 8. It doesn’t matter how much or where you make spike protein we’ve got antibodies schmantibodies 9. It’s no more contaminated than any other vaccine 10. There was no bait and switch 11. We know how to analyze and test all the important components 12. It’s safe and effective and of high quality