reSee.it - Tweets Saved By @IamBrookJackson

@IamBrookJackson - Brook Jackson 💜
BREAKING: Commit Fraud in a Clinical Trial? You Will Go to Jail...unless you are Pfizer! 🔴Data falsification: Trial results were fabricated undermining the entire drug approval process. ⚠️ FDA was misled: The agency was fed false data that could have shaped regulatory decisions. 💊 Health risk: These trials involved asthma drugs used by millions. 💰 Motive? Money. Like always. But unlike the small fish who get charged, Pfizer walks away untouched—again. https://ourcommunitymedia.org/us-news/owners-of-clinical-research-facility-plead-guilty-to-fraud-in-asthma-drug-trials/


@IamBrookJackson - Brook Jackson 💜
Call it a beacon and a battleground, David v Goliath, a beautiful disaster maybe...idk, but to me this case offers a glimmer of hope that justice might still be possible. It's already created an unerasable record of facts that expose a depth of corporate fraud & government collusion on a scale not many can fathom. It's tragic in the ruthless effort to bury it and me, but most of all in the complete disregard for the injuries & lives that have been shattered. Justice demands accountability-and I'm here for it! Below is our request for a 60-day extension to file our brief. We were granted 30, so I won't complain mainly because this is so damn good: "Relator Brook Jackson alleged that Defendants Pfizer Inc., ICON PLC and Ventavia defrauded the government by claiming federal funds to deliver COVID-19 vaccines authorized or approved by the FDA for the safe and effective prevention of COVID-19. She alleged that Defendants engaged in fraud in the design, conduct, analysis, and reporting of clinical trials to obtain Emergency Use Authorization (EUA) for a vaccine against COVID-19. Overwhelming evidence developed without formal discovery established that defendants knew the modified RNA technology developed by defendants and marketed by Pfizer did not prevent infection or transmission of SARS-Cov-2; that Pfizer’s vaccines posed unreasonable risks of serious harm to injected individuals; and that defendants’ clinical trial fraud was necessary to obtain authorization under the objective standards of the EUA statute, 21 U.S.C. § 360bbb-3. Relator seeks return to the federal fisc treble damages and penalties for the billions of dollars wrongfully paid to Pfizer under its contracts with the government. This appeal is from the District Court’s order entered on August 9, 2024, granting the United States Department of Justice’s Motion to Intervene and Dismiss Relator’s qui tam action against Defendants under 31 U.S.C. § 3730(c)(2)(A), and granting Ventavia Research Group, LLC’s Motion to Dismiss Relator’s employment retaliation claim. Both motions were extensively briefed by Relator and the 25-page order was entered by the District Court more than three months after a two-and-a-half hour hearing held on May 1, 2024. Relator anticipates that preparation of Appellant’s Brief will require an extensive and coordinated effort to conduct a detailed review of the briefing and hearing transcript leading up to the issuance of the order. The Department of Justice’s motion raises important questions under the United States Constitution and its Amendments, the False Claims Act, and the Federal Rules of Civil Procedure. Many case authorities were addressed in the briefing before the District Court and substantial additional research will be required of Appellant’s counsel to update developments in this novel area of law. In addition, Relator’s complaint and the opposition to the motion present several complex factual issues concerning the scientific fraud of which defendants were accused, and the ultimate consequences to public health from authorization of a vaccine with negative efficacy and causing serious harm. Relator’s opposition to the Department of Justice’s motion included declarations by Dr. Peter McCullough and Joseph Fraiman, and thirteen exhibits, consisting of scientific literature, court filings and correspondence between the Department and Senator Charles Grassley, the author and principal protector of the False Claims Act. ‼️Because this appeal involves an unprecedented motion by the United States Department of Justice, based on a contested statement of national health policy, to intervene for purposes of dismissing an action shown (without evidentiary contradiction) to be meritorious, Relator anticipates an unusual amount of work will be required to prepare the opening brief. ‼️ Relator and her counsel intend to review the pertinent case law, from before and after the Supreme Court’s decision in United States ex rel. Polansky v. Exec. Health Res., Inc., 143 S. Ct. 1720, 1734 (2023), the extensive legislative history of the False Claims Act and its amendments, and the changing government statements of national health policy in relation to the now widely recognized fraud by defendants in developing the modified genetic biologic vaccine technology." See ya in court, fellas. Happy holidays😘

@IamBrookJackson - Brook Jackson 💜
‼️ 623 pages explains why the U.S. DOJ demands my whistleblower case against Pfizer be dismissed. The judge in our case must ensure that justice is served! Does he have the diligence and integrity? We await his decision. https://judiciary.house.gov/sites/evo-subsites/republicans-judiciary.house.gov/files/evo-media-document/2024-06-24%20Politics%20Private%20Interests%20and%20the%20Biden%20Administrations%20Deviation%20from%20Agency%20Regulations%20in%20the%20COVID-19%20Pandemic.pdf https://t.co/YqbwI5AYjq

@IamBrookJackson - Brook Jackson 💜
UPDATE: We just filed a Notice of Supplemental Authority which supports our opposition to DOJ’s Motion to Intervene & Dismiss. 1. United States v. Academy Mortgage Corp. The govt must show “good cause” to intervene/dismiss. Since Pfizer failed to get the job done, DOJ now claims “because we said to” is good enough cause. It’s not.
@IamBrookJackson - Brook Jackson 💜
2. National Rifle Association of America v. Vullo SCOTUS held that govt officials cannot use their power selectively to punish or suppress speech based on viewpoint discrimination. DOJ’s motion is based on my viewpoint regarding Pfizer’s clinical trial practices & the safety/efficacy of their product, which constitutes this type of discrimination. Haters.
@IamBrookJackson - Brook Jackson 💜
3. Federal Drug Administration v. Alliance for Hippocratic Medicine There are no other effective avenues for redress against the FDA’s actions regarding Pfizer’s EUA. This underscores the need for rigorous judicial oversight, protection of constitutional rights, and the role of the FCA in allowing private individuals to bring actions on behalf of the govt. I still believe!❤️
@IamBrookJackson - Brook Jackson 💜
https://www.iambrookjackson.com/_files/ugd/9df0bc_c98865d557094e169877cd7323c88967.pdf

@IamBrookJackson - Brook Jackson 💜
🚨NEW: One of the most powerful legal briefs I have ever read. To my team of attorneys — thank you for your tireless efforts, your patience, your commitment to excellence and the rule of law. The principles of humanity have been evident in every action you've taken, ensuring that justice is served with integrity. I totally admire that! RELATOR’S SUR-REPLY TO GOVERNMENT’S LATE REPLY RE MOTION TO INTERVENE AND DISMISS TO THE HONORABLE JUDGE OF THE COURT: Relator Brook Jackson (“Relator”) hereby requests that the Court consider this sur-reply to the late reply of the United States of America (“Government” or “DOJ”) to Relator’s opposition to the DOJ’s motion to intervene to dismiss. INTRODUCTION In its effort to gain permission to intervene and dismiss this extraordinarily important False Claims Act case, the DOJ asserts several misleading half-truths. Yes, Brook Jackson’s qui tam action is brought in the name of the Government and the United States is the real party in interest. But, in making the False Claims Act “the Government’s primary litigative tool for combating fraud” “in modern times,” Congress partially assigned rights directly to relators like Brook Jackson, equipping them with authority to litigate qui tam claims without control or even direct participation of the DOJ. Brook Jackson does not need approval of the DOJ or agency executives to fulfill her role before this Court.
@IamBrookJackson - Brook Jackson 💜
Yes, Congress provided in 31 U.S.C. § 3730(c)(3) a mechanism for the Government to file a motion seeking dismissal of the qui tam action notwithstanding a relator’s objections. But, as the Supreme Court made clear, the Government first must show good cause to intervene to become a party to the action, and even then, the Government must “offer[] a reasonable argument for why the burdens of continued litigation outweigh its benefits.” Congress intended that False Claims Act claims be dismissed for legitimate government purposes, and not as a result of fraud, illegality, or lack of political will. And yes, the courts have, both before and after Polansky, found “good cause” for intervention under § 3730(c)(3) based on the Government’s reason for moving to dismiss under § 3730(c)(2)(A), “itself.” But in each and every case, the Government was able to show a reasoned basis for intervention and dismissal, grounded in a valid governmental purpose as exemplified in the Granston Memo. Never before has the DOJ sought to dismiss a meritorious case like Brook Jackson’s case here, where the relator can demonstrate a strong basis to recover substantial damages for harms caused to the United States by fraud and false claims on the public fisc. And, never before has the Government failed to even articulate a legitimate reason for dismissal.
@IamBrookJackson - Brook Jackson 💜
In its late reply, the DOJ waives key points and authorities raised in the opposition. The catastrophic harms caused by Pfizer’s ineffective and unsafe modified RNA biologic, the loss of credibility in agencies and the DOJ as they abdicate public policy to partner up with corporate interests, and the essential role courts hold keeping our Government functioning in the face of executive failures, are all now undisputed. The DOJ continues to assert an unfettered right to dismiss any qui tam action whenever it wants, without mention of the Granston Memo. It offers no explanation for how this lawsuit seeking to hold Pfizer accountable for proven fraud in the design, conduct, analysis and reporting of the clinical trials is contrary to national health policy. The Government does not deny that dismissal would undermine the Act by sending a chilling signal to future relators that exposing fraud on the United States is futile when agency executives want that fraud kept secret. Nor does it deny that this is its true motive for seeking dismissal. The DOJ does not address Rule 24 of the Federal Rules of Civil Procedure, or the requirement under subdivision (d)(3) of that Rule that the Court consider prejudice to Brook Jackson as the original party in determining whether to grant permissive intervention. And, it completely ignores constitutional arguments regarding content-based infringement of Brook Jackson’s First Amendment right to petition, the disordering of the separation of powers to protect corporate and executive actors, and the lack of a rational basis under due process and equal protection. While the Government possesses the authority, indeed obligation, to make a “later date” intervention to dismiss qui tam actions on good grounds based on legitimate government purposes consistent with the False Claims Act, no such showing is made here. The DOJ’s motion should be denied without prejudice on this record. In the event that the Government seeks to intervene post-seal period based on actual evidence and articulated grounds, the Court should allow limited discovery and hold an evidentiary hearing under § 3730(c)(2)(A) and (c)(3).
@IamBrookJackson - Brook Jackson 💜
SUR-REPLY ARGUMENTS I. The DOJ Failed to Show Good Cause for Intervention Contrary to the DOJ’s contention, no court has ever relieved the DOJ of the obligation to make a showing of good cause for a “later date” intervention under § 3730(c)(3). Every court which has found good cause to intervene based on the Government’s motion to dismiss did so because the Government had a reasonable basis for seeking dismissal: i.e., the Government “offer[ed] a reasonable argument for why the burdens of continued litigation outweigh its benefits.” Polansky., 143 S. Ct. at 1734. And, since the Supreme Court in Polansky rejected the DOJ’s assertion of unfettered discretion to dismiss, no Court has held that the government’s mere assertion of the motion, itself, satisfied good cause. In each, it was the substantive reason for the dismissal, itself, that provided good cause. The assertion of an unfettered discretion here fails to provide good cause. Polansky provides a perfect example. In discussing the good cause finding by the Third Circuit, the Supreme Court stated the actual grounds for seeking dismissal, not the mere assertion of the unfettered right, as showing good cause. And applying that standard, the Third Circuit found that the Government’s request to dismiss the suit—based on its weighing of discovery burdens against likelihood of success—itself established good cause to intervene. As pointed out in the opposition but ignored in the reply, these reasons for dismissal included evidence that actual “discovery burdens mounted and weighty privilege issues emerged,” and the Government had “thoroughly investigated the cost and benefits of allowing [Polansky’s] case to proceed and ha[d] come to a valid conclusion based on the results of its investigation.” 143 S. Ct. at 1729 (emphasis supplied). “Polansky [did] not challenge that conclusion.” No such showing is made by the DOJ here.
@IamBrookJackson - Brook Jackson 💜
Similarly, the DOJ ignores the actual basis for dismissal established in Brutus Trading, LLC v. Standard Chtd. Bank, 2023 U.S. App. LEXIS 21868, at *5 (2d Cir. Aug. 21, 2023). There, the Government showed that the relator's “factual allegations were unsupported, its legal theory was not cognizable, and the continuation of the suit would waste considerable government resources.” Contrary to the DOJ’s position here, it was not the mere assertion of an unfettered right to dismiss that established good cause. In United States ex rel. Carver v. Physicians Pain Specialists of Ala., P.C., 2023 U.S. App. LEXIS 19592, at *11-13 (11th Cir. July 31, 2023), it was the “same grounds” supporting dismissal that also established good cause, not the mere assertion of a right. (Emphasis supplied.) Those grounds included evidence that Carver had “failed to prosecute this action to an enforceable judgment, neglected her responsibilities as a relator, burdened the United States with discovery requests that are either irrelevant or premature, and undercut the United States’ FCA enforcement efforts in this district.” Id. The DOJ in Carver provided great detail for these considerations, showing that “it did not make the decision ‘lightly,’ [and] it had ‘determined that the costs of continued litigation outweigh any benefits the United States could realistically obtain.’” No such claims could be made in connection with Brook Jackson’s lawsuit here. In United States. ex rel. USN4U, LLC v. Wolf Creek Fed. Servs., 2023 U.S. Dist. LEXIS 217620, at *4-5 (N.D. Ohio Dec. 7, 2023), the district court expressly found that the Government had “shown good cause to intervene.” In Wolf Creek, the district court held: Here, for good cause the U.S. contends that discovery has cast doubt on the Relator's ability to prove any False Claims Act violations against Defendants. Many of the Relator's allegations and his expert's opinions have been challenged by the testimony of the NASA employees who were deposed in this case. For example, Relator argues that NASA employees did not adequately review Defendants' proposals, but the NASA employees described a lengthy review process for the approval of the proposals. And the U.S. correctly questions the ability of Relator's expert to refute this because he was not involved in NASA's review process. See ECF Doc. 69-1 at 7. The U.S. also shares the concerns of [*5] the Court regarding Relator's credibility; his testimony during the October 4, 2023 hearing was "vague, evasive and contradictory." Id. The U.S. does not want to devote any more resources to the case given the unlikelihood of Relator's success. [Id.] The district courts decision is currently being appealed, mainly on other grounds. But even this ruling supports Brook Jackson here, as the DOJ fails to point to any of the factors that were found to be present there.
@IamBrookJackson - Brook Jackson 💜
The DOJ disingenuously accuses Relator of asserting that courts “look beyond the flexible ‘good cause’ standard.” In fact, Relator acknowledges and embraces the good cause standard. Despite its flexible, familia and non-burdensome nature, the Government simply does not meet it. Similarly, the DOJ claims that it is not required to ground its “later date” intervention on “new evidence” or “changed circumstances,” but it cites no authority in that regard. In contrast, Relator in her opposition quotes the Supreme Court’s decision in Polansky explaining Congress’s purpose for “good cause” later date permissive intervention, because it “knew circumstances could change and new information could come to light.” Polansky, 143 S. Ct. at 1733. In sum, none of the courts since Polansky have agreed that the DOJ has unfettered authority to intervene to dismiss qui tam actions. Neither the language nor the purpose of the qui tam provisions support the government assertion of that position here.
@IamBrookJackson - Brook Jackson 💜
II. The DOJ Failed to Address Brook Jackson’s Constitutional Arguments The DOJ makes little effort to address the detailed constitutional arguments presented in the opposition. In order to evade its obligation to act consistent with constitutional constraints, the DOJ suggests that Brook Jackson may petition the government elsewhere – through some “other appropriate avenues” – to raise “concerns about federal agency decision-making.” The Government does not deny that Congress partially assigned to Relator the right to petition the Government for redress of the Goverenment’s injury. Nor does the Government refute that it would be a violation of the First Amendment to infringe on Brook Jackson’s speech in the public square based upon her viewpoint that Pfizer committed clinical trial fraud to the injury of the United States and its People. And thus, the DOJ offers no explanation for how this content-based restriction of her right to petition under the qui tam statute complies with the First Amendment.
@IamBrookJackson - Brook Jackson 💜
Worse, the DOJ completely ignores the point raised in the opposition that separation of powers concerns must weigh heavily in the “good cause” determination. Here, the DOJ’s motion to dismiss should be carefully and independently reviewed by this Article III Court, as the Government attempts to rewrite both the objective standards in EUA statute and the 1986 False Claims Act amendments which eliminated the “government knowledge” bar. Government knowledge, acquiescence or complicity in Pfizer’s clinical trial fraud would not excuse the sponsor of its obligations to conduct non-fraudulent trials on its biologic product. In these circumstances, the Court is the last bastion of hope for a Government that adheres to the Rule of Law. Finally, the DOJ does not dispute, and therefore concedes, that substantive due process and equal protection require that its motion have a rational basis. The Government’s abject failure to make a coherent explanation for dismissal means it fails even this minimal test. There can be no doubt that protection of whistleblowers is a principal policy of the United States. So too is prevention of fraud in the design, conduct, analysis and reporting of clinical trials. Given this, the DOJ is unable to explain to the Court, or the People, why Brook Jackson’s prosecution of this action is inconsistent with national policy. Rather, dismissal of this case, would be.
@IamBrookJackson - Brook Jackson 💜
CONCLUSION The Court should grant leave to file this sur-reply, and deny the DOJ’s motion to intervene and dismiss for the reasons stated herein, in the opposition and at oral argument. Respectfully 💜
@IamBrookJackson - Brook Jackson 💜
@threadreaderapp unroll

@IamBrookJackson - Brook Jackson 💜
🚨CONFIRMED: The United States & Pfizer orchestrated a scheme, exploiting emergency use powers delegated by Congress to Secretary of HHS, Xavier Becerra. This evil & calculated maneuver aimed to deceive the entire world's population, fostering a false belief that COVID-19 shots, allegedly proven safe & effective, had undergone rigorous & well-controlled clinical trials. THIS IS A LIE. Their deceptive & manipulative approach in the dissemination of information about the products was intentional, fraudulent & treasonous. An immediate & uncompromising recall of these falsely labeled & contaminated products is non-negotiable. In addition, we should unequivocally demand that Congress initiate immediate hearings to address matters of public concern & safety. Congress must fulfill its duty to investigate & address these matters without delay. https://x.com/phillyharper/status/1737804438956323180?s=20

@IamBrookJackson - Brook Jackson 💜
🚨The World vs Pfizer (case update) “The right to search for truth implies also a duty; one must not conceal any part of what one has recognized to be true.” - Albert Einstein As indicated by the quote above, inherent in the scientific search for truth is a duty to disclose, and not conceal, any part of what is shown to be true. Relator Jackson alleged Pfizer abandoned the scientific method and the search for truth to induce FDA’s authorization of its modRNA biologic to enable it to sell the United States 100M doses (for $1.9B). Ms. Jackson has alleged Pfizer engaged in fraud in the design, conduct, analysis and reporting of its clinical trials, and in statements it made to FDA.

@IamBrookJackson - Brook Jackson 💜
A statement from the U.S. that it has full confidence in Pfizer’s “vaccine” is one more red flag and a sign that there is a f*cking problem. @pfizer - you don’t get to speak for The People, I do. There is no burden on us to begin the discovery process. You’re just terrified! https://t.co/1Luk0eMhWk

@IamBrookJackson - Brook Jackson 💜
🚨I am suing Pfizer again. 1. Coercion of participants 2. Lack of informed consent 3. Data manipulation & fraud 4. Hidden severe adverse events (tethered spines & fetal deaths in RSV) 5. IRB fraud https://www.blast-info.fr/articles/2023/vaccin-contre-la-bronchiolite-pfizer-essais-en-zones-dombre-SM_0QNVMT76A_KFpDed6BA

@IamBrookJackson - Brook Jackson 💜
🚨I am suing Pfizer again. 1. Coercion of participants 2. Lack of informed consent 3. Data manipulation & fraud 4. Hidden severe adverse events (tethered spines & fetal deaths in RSV) 5. IRB fraud https://www.blast-info.fr/articles/2023/vaccin-contre-la-bronchiolite-pfizer-essais-en-zones-dombre-SM_0QNVMT76A_KFpDed6BA
@AMK_PhD - Ahmad M Khalil 🇺🇸
@IamBrookJackson First lawsuit still pending?!
@IamBrookJackson - Brook Jackson 💜
@AMK_PhD Yep. Our response to Pfizer’s motion to dismiss due Dec. 1.

@IamBrookJackson - Brook Jackson 💜
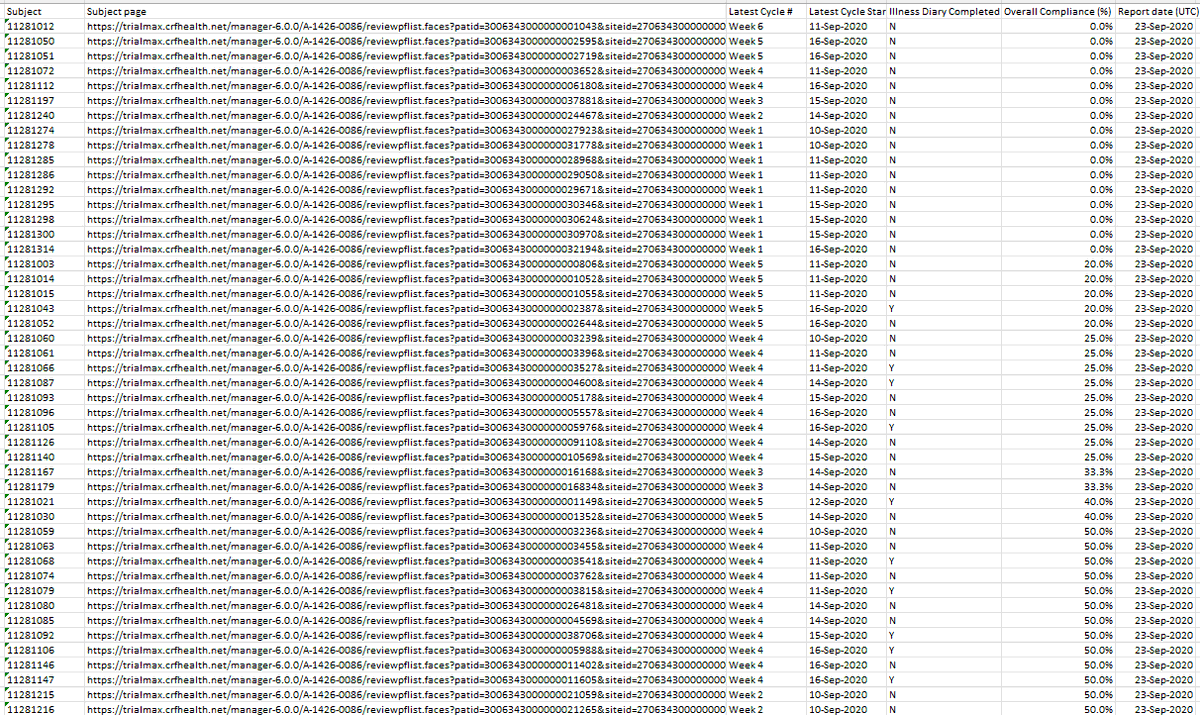
What I can also confirm, @TheJusticeDept, is that ALL clinical trial sites had compliance issues with the eDiary; NOT recording adverse events and NOT completing illness diaries. *see Pfizer protocol amendment 8, dated 15Oct2020 https://t.co/AdtuYS0svh

@IamBrookJackson - Brook Jackson 💜
Pfizer: Reports of lymphadenopathy were imbalanced with notably more cases in the Comirnaty group (87) vs placebo group (8). FDA: Remove that part, bro. Pfizer: Okay, there were NO OTHER NOTABLE patters or NUMERICAL IMBALANCES between the two group. https://t.co/LRPgKVKGo3
@IamBrookJackson - Brook Jackson 💜

@IamBrookJackson - Brook Jackson 💜
MIT engineers design more powerful RNA vaccines—“with intranasal vaccination, you might be able to kill Covid (SARS-CoV-2) at the mucus membrane before it gets into your body.” https://news.mit.edu/2023/mit-engineers-design-more-powerful-rna-vaccines-0907 https://nature.com/articles/s41551-023-01082-6


@IamBrookJackson - Brook Jackson 💜
Objective: To provide prevention of Covid-19 infection for weeks or months with a single injection of a long-acting formulation of ivermectin. Funded by: Bill n’ Melinda Gates Foundation @pfizer Ivermectin. Because it works. https://medincell.com/en/2023/01/05/medincell-announces-positive-results-for-the-saive-clinical-study-in-prevention-of-covid-19-infection-in-a-contact-based-population/