TruthArchive.ai - Tweets Saved By @a_nineties

@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
🚨🚨FINAL CLINICAL STUDY REPORTS FOR ASTRAZENECA🚨🚨 https://mega.nz/folder/OJwTgazL#pYVUqu2y3jUkWLFttRD9bQ… 51 documents - 350 MB - released on EMA CDP 26.4.2024 🧐🧐 sketchy timing re: withdrawal. haven't read anything yet, directly reuploaded @eh_den @ClareCraigPath

@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
@eh_den @ClareCraigPath just browsing through the safety summary at work. healthcare providers are familiar with anaphylaxis soooo it's not worth bothering with. OH and all the excipients you might be allergic to are of course known and communicated to you so you can make an informed decision 🤡🤡
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
no TTP "reported" during the trial but "very rarely observed" in post-marketing - MoA is "unconfirmed hypothesis" - and a whole 'nother bunch of subjects for whom "the benefits and risks of vaccination should be considered". very unusual tones for a vaccine company 😊
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
look at the dates on the cited studies 😂😂 this section is especially egregious because AZD1222 had an eye-wateringly enormous GBS signal.
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
this is what happens when you outsource pharmacovigilance to the pharmaceutical company 😅i wouldn't bother CONFIRMING any of those reports either. why even list them at all if they're not CONFIRMED? isn't serious disease CONFIRMATION enough? infuriating
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
😂you'll be pleased to hear AZD1222 has a slight positive effect on all-cause mortality according to this summary analysis and saline injections have twice the related AESI rate. AZ didn't exclude subjects who received non-study covid vaccines in the c00001 trial..same here?
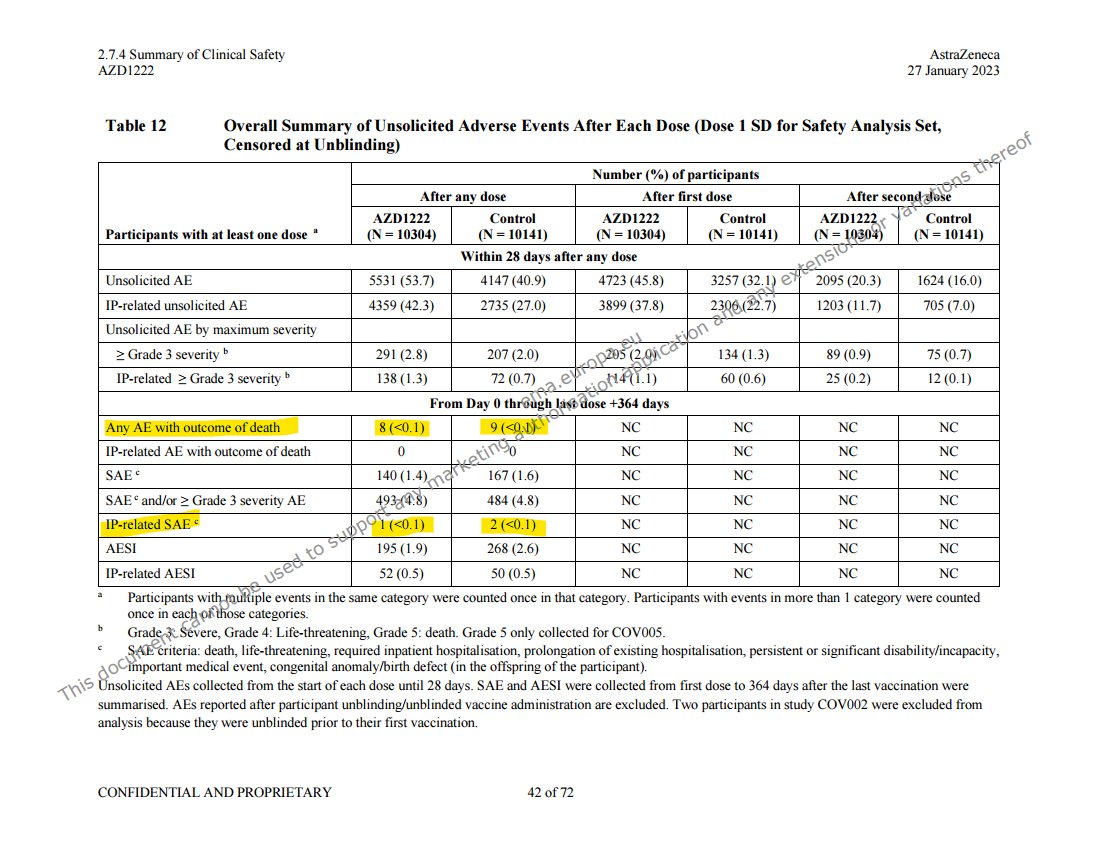
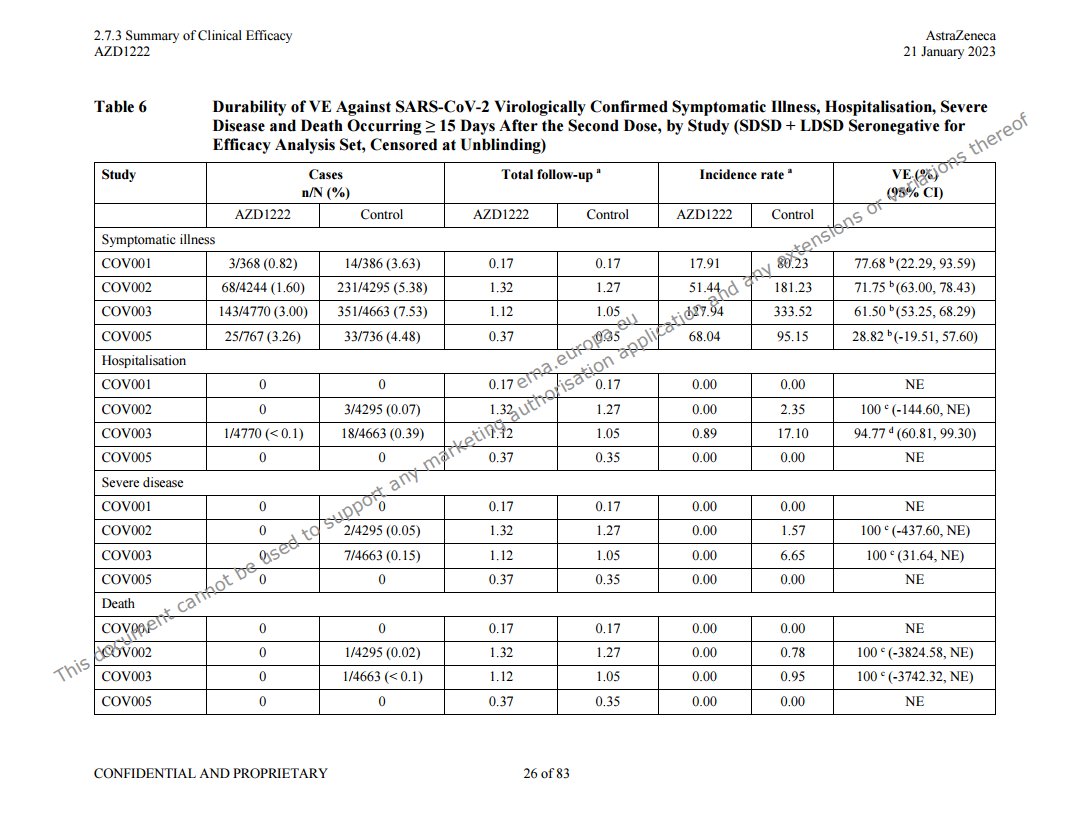
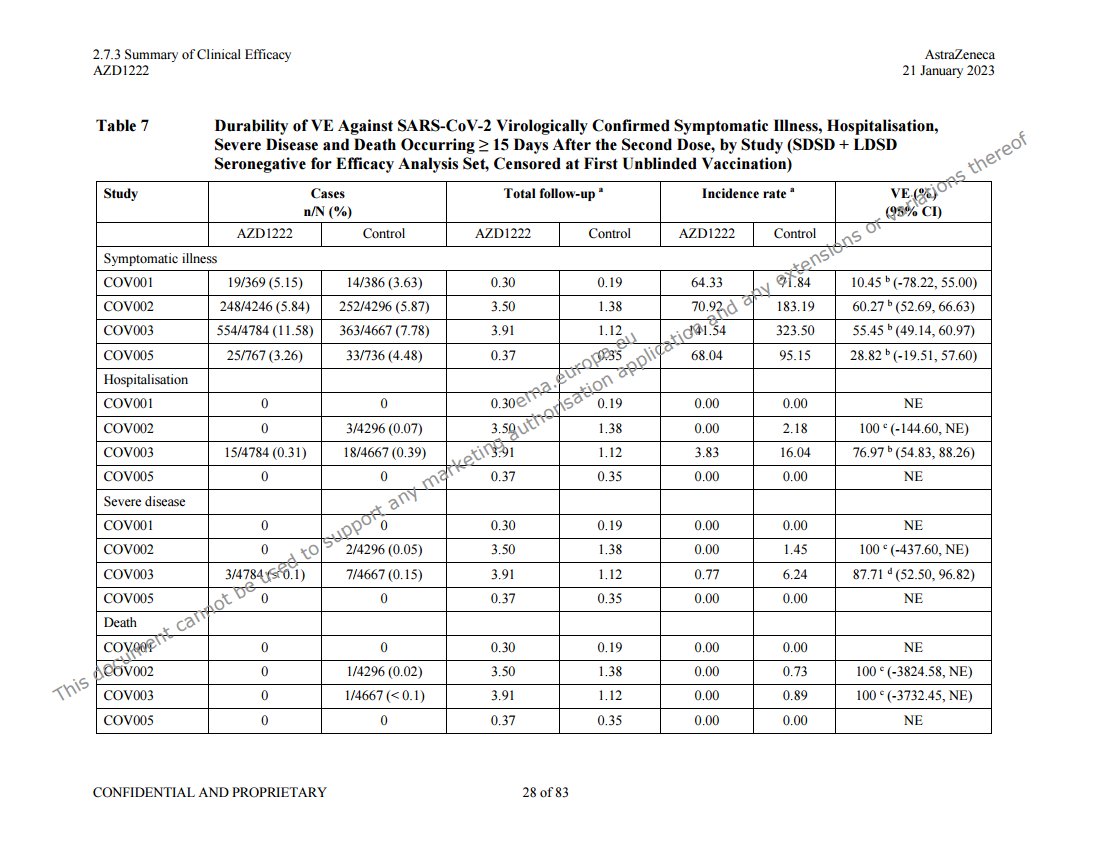
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
these two tables are right after each other in the efficacy summary file, p28. https://mega.nz/folder/OJwTgazL#pYVUqu2y3jUkWLFttRD9bQ/file/bZgwSZyQ "first unblinded vaccination" means vaccination in or out of the study, so it's a later timepoint than "unblinded", non-AZ covid jabs weren't excluded. pretty bad either way!


@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
the very first goddamn email: daszak says baric is working on nanoparticles, and baric is asked about sars glycoproteins he's making for a DARPA grant and they're trying to get them into bats from march 2018
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
jesus christ, "batized" mice, as in mice frankensteined into having a bats immune system?
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
who the hell is tonie rocke and her plan to contract a company to build a bat spraying device? i also find the reference to "quid pro quo" lab work a little concerning - no paper trail is what makes it easier than $
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
i'd also like to draw attention to this post i found very insightful from the original thread in the OP, comment section is very worth reading
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
deviant spike proteins being developed https://t.co/SBDVcn0Dsr

@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
🚨🚨NEW DNAse FOIAs!🚨🚨 i just got into contact with the recipient of the original two documents in the quote tweet. he received these two new ones on 6.12. 1)https://mega.nz/file/GIolzLDZ#Y-2dlSbF4VfA28S1mjJm_CCcn7mtTlOiHUBUO5AYhaU… 2)https://mega.nz/file/eIoSyQyS#Y2g4VtSCUlDXg9JSTY32BQIM8_Up9I8xtrkg0SMxIFw… @DJSpeicher @CanningPharm @JesslovesMJK @Kevin_McKernan


@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
document 1: 17.8.2021. 9 pages spec = "tell us whatcha got LOL" andover = partially cleared rec#3 +7= still uncleared https://t.co/Rl16LCDh1K
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
GMP-inspection= NOT SATISFACTORY. tick is in the wrong column, pr brigade pharma bootlickers! https://t.co/77gPFQDfah
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
the on-site andover tests used to validate the enzymes used return results in different units than the vendor certificate of analysis! the vendors check their stuff before sending to pfizer, but andover uses different measures?? please correct me if im wrong https://t.co/lPv4FF4dPl
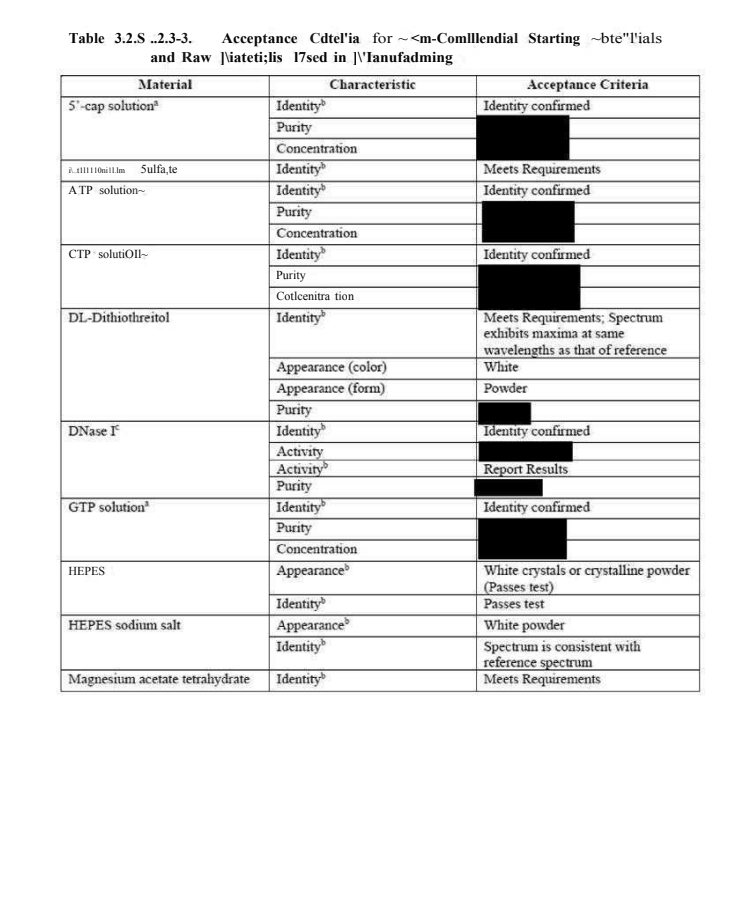
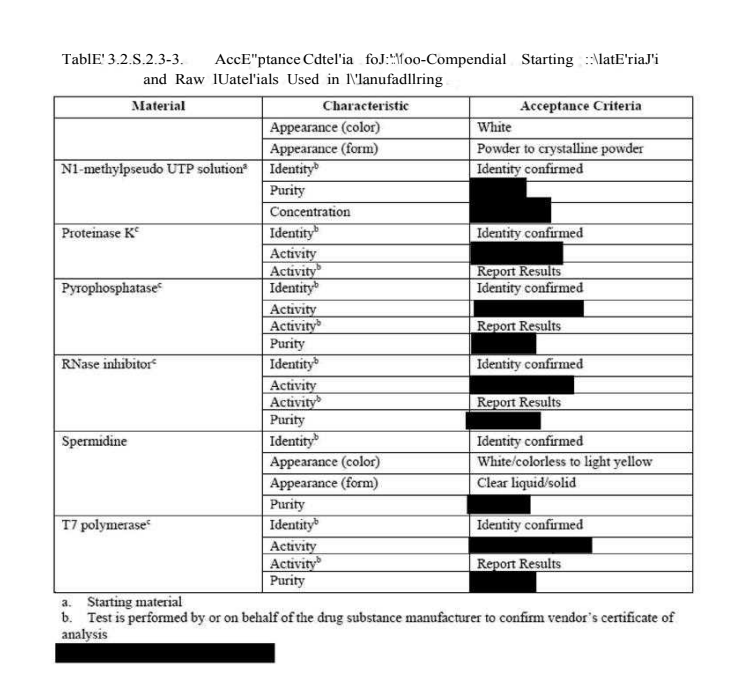
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
check out that garbled heading, wtf, and then the 35 dpi scan. i think most of the black boxes are "report results" following from what the previous page said. https://t.co/ngEeKh2Avn
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
reminder: this document is AUGUST. they want studies by end of Q2, that was 1.5 months ago. and then the red boxed paragraph! i will have to quote my meme. dont worry though! pfizer is rawhiding it by just measuring DS! https://t.co/zLxw6WbFDU https://t.co/1w6YDsjcmX
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
THE EMA IS PFIZER'S BITCH -no descriptions of tests -no vendor data validation -"pls jus gib short description of good-enough result to poor regulator" aka my boss is killing me :((( -made experiment(S) -wont gib data -"gib jus 1 studY pls" -but after approval https://t.co/Cxx5V8oT8k
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
no obligations - just recommendations. extension requested to q2 2021. nothing doing, no dice, nix nada niente by mid august. so EMA folds and "recommends" they "follow-up". but it's enough for partially fulfilled, and that's enough for european arms. https://t.co/FQB5N8sXoT
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
nine months after the jabs hit the market, the EMA gave up on getting the data on process 2 quality control, especially regarding ALL the enzymes used in creating the DNA template and the efficacy of the DNAse treatment! https://t.co/KwanNLa3PA
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
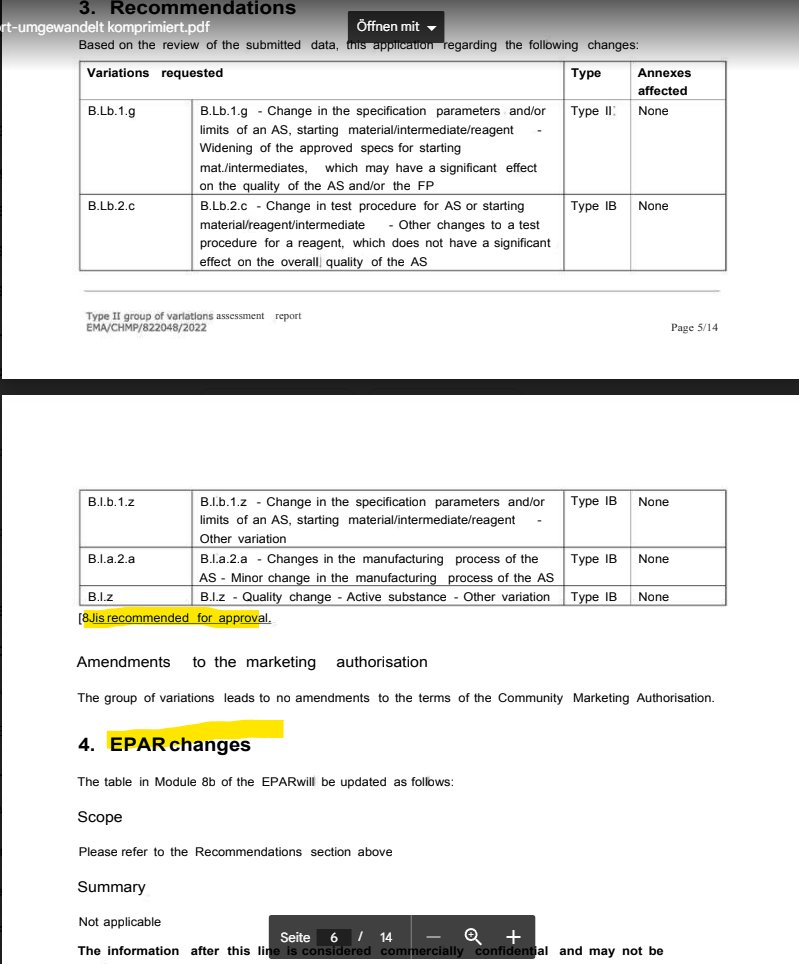
second document: 8.12.2022. just about a year ago, that's like yesterday by FOIA standards! this one is 14 pages, but the table of contents looks pretty weird, like some of the headings in the previous file 🤨 https://t.co/QaxRwmvcFF
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
1. spec weakened 2. spec will be introduced (is not active yet) 3. spec removed 4. describe spec (is not yet described) 5. figured out how much the filters can take (didnt know that yet) 6. REC#7 is fulfilled???? https://t.co/VHvDu3vDTz
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
nope hold on, both RECs #3 and #7 are just gucci now. how come?! new results for enzyme testing, so lets change the specs we had so far at andover, and apply those to mainz and marburg SO THEY FINALLY HAVE SOME TOO. december 2022! riSk-bALaNcE rEmAiNs PoSiTivE!!11! https://t.co/W0kvMDPA64
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
the table just says the previous changes are to be approved. then come the EPAR changes, guess the plebs should be informed, eh? first comes a long version of what was said previously, with some extra detail on how cucked the EMA is to approve this. https://t.co/w5aiwvfHu9
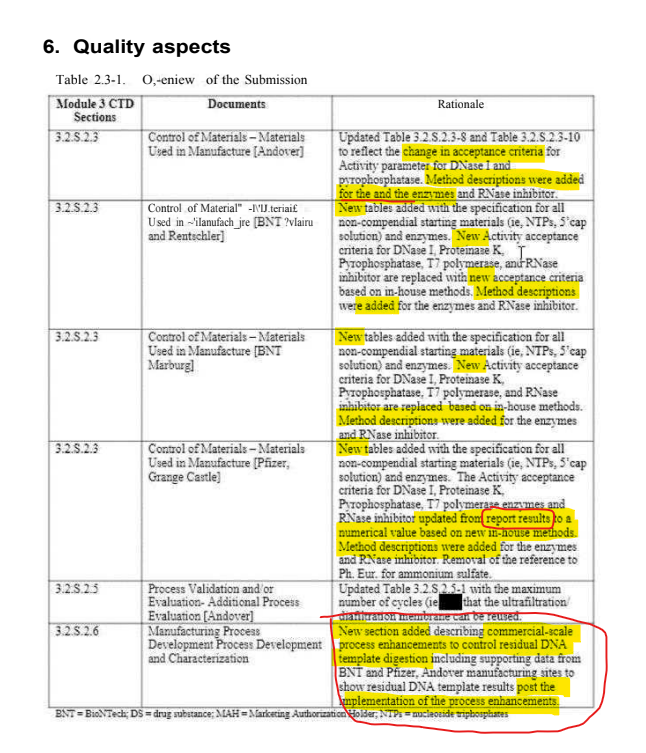
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
wow! "for the and the enzymes" lil' mistake there oopsie, everything so nice and new. the last box is the absolute kicker. COMMERCIAL-SCALE PROCEES ENHANCEMENTS WER IMPLEMENTED BY DECEMBER 2022. everything prior to that was filthy, pfizer just refused to share the dirty data! https://t.co/sEOytCBDAN
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
it's really astonishing. they were running mainz and marburg for over 1,5 years without a shred of data on enzyme activity at those sites. once commercial process enhancements were implemented by december 2022, they took these new specs for all sites.
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
pfizer just told regulators to get fucked for 18 months. then they had a new process they could probably game better and the relieveeeeeeeeed regulators were oh so fucking glad to finally say "ah yes this looks fine". it's insane. i say that often but DAMN
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
dont know when the previous variation was (can look it up tho i think) but thats when ANDOVER got specs, probably after implementing "process enhancements". pfizer was then asked and AGREED to apply these to the kraut sites. and all this crap for months for +/- 3 standard devs! https://t.co/yEa2BYCrev
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
the DNAse was way too weak, and the pyrophosphatase too "acitve"? and look at rentschler just appear out of nowhere as a german DS site, previously only marburg and mainz were mentioned. https://t.co/O91UJn0dy0
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
what would a foia without a few wiped pages be? the last bit before the redaction is important but forgot to highlight it. "No new small-scale experiments are planned or considered to be needed to show correlation between DNase I activity and levels of residual DNA." https://t.co/xxjtu1Fij1
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
it ends with some bureaucratic reminder. so PFE/BNTX flew blind for 18 months under the watchful eyes of EVERY REGULATOR, didnt deliver SHIT, then "implement process enhancements", measure less somehow, that's it we're done here close the file. https://t.co/8haSLpny01

@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
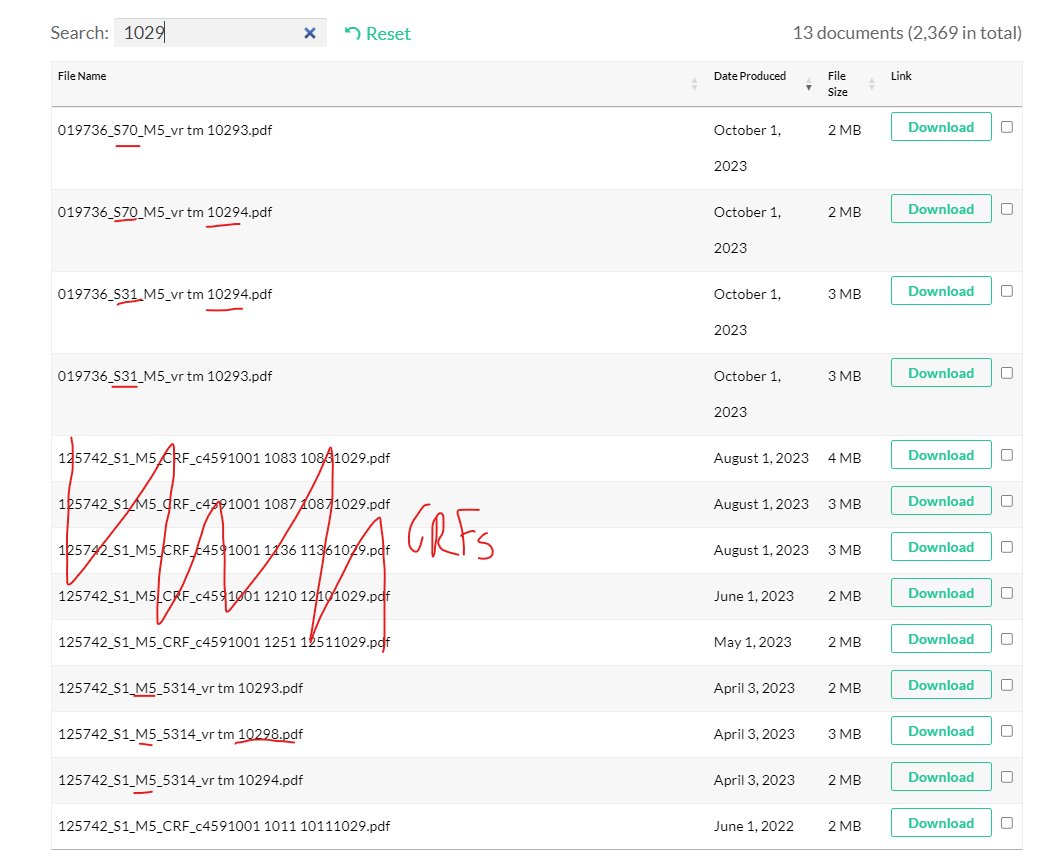
while scrolling through the october documents i spotted this little coof strain sequencing paper VR VTN 10436 i had somehow missed. now a lot of it is pretty beyond me, but one thing really made me go "hmm" https://phmpt.org/wp-content/uploads/2023/10/125742_S6_M5_5314_vr-vtn-10436.pdf
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
"Midturbinate swabs that are positive in the SARS-CoV-2 RT-PCR (Cepheid) assay at either the N or E gene target are advanced for viral sequence." these are the swabs from C4591001 trial participants. https://t.co/ihaafqwWDq
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
why are swabs chosen by gene detected instead of by ct value? why dont they sequence the spike-only probes? what am i missing here? is the assumption that non-N&E are just fragments? https://t.co/J8VExDMEC6
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
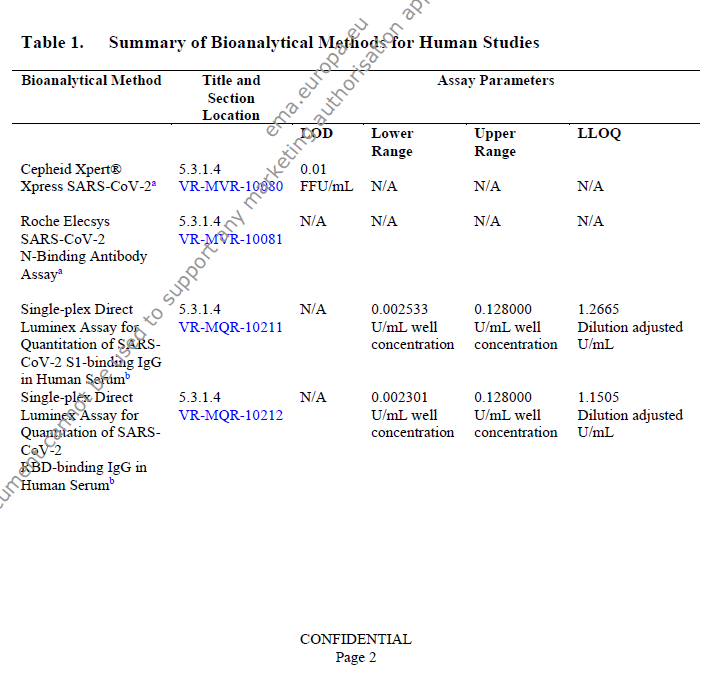
for anyone versed in this stuff or otherwise interested, here's the assays validation https://phmpt.org/wp-content/uploads/2023/04/125742_S1_M5_5314_vr-mvr-10080.pdf the 10076 file doesn't seem to be on PHMPT however, and the other two 10080 documents are entirely redacted. https://t.co/fqVuzp6Ymo
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
if they're looking for spike mutations it might be smart not to choose spikes that match a wuhan-1 pcr, it just occurred to me^^ that also explains the either N or E gene choice. they're missing out on the N & E mutants tho? 🧐
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
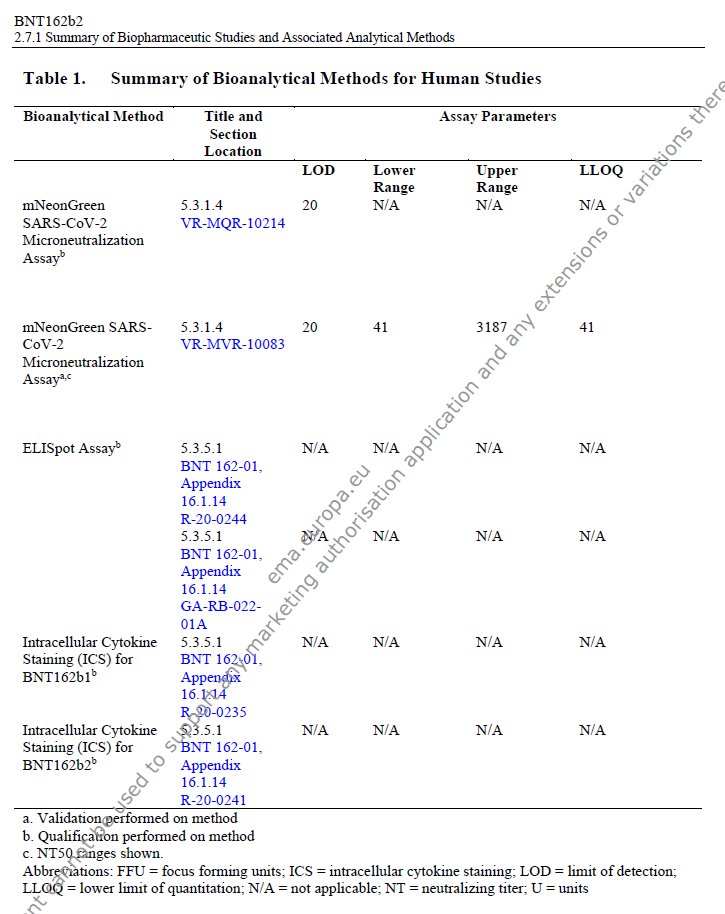
there's no 10295 on phmpt either, but we have: 3x 10293 IgG S1 serum ab luminex assay 3x 10294 IgG RBD luminex 1x 10298 virus microneutralization assay https://t.co/B17H1jZ0nG

@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
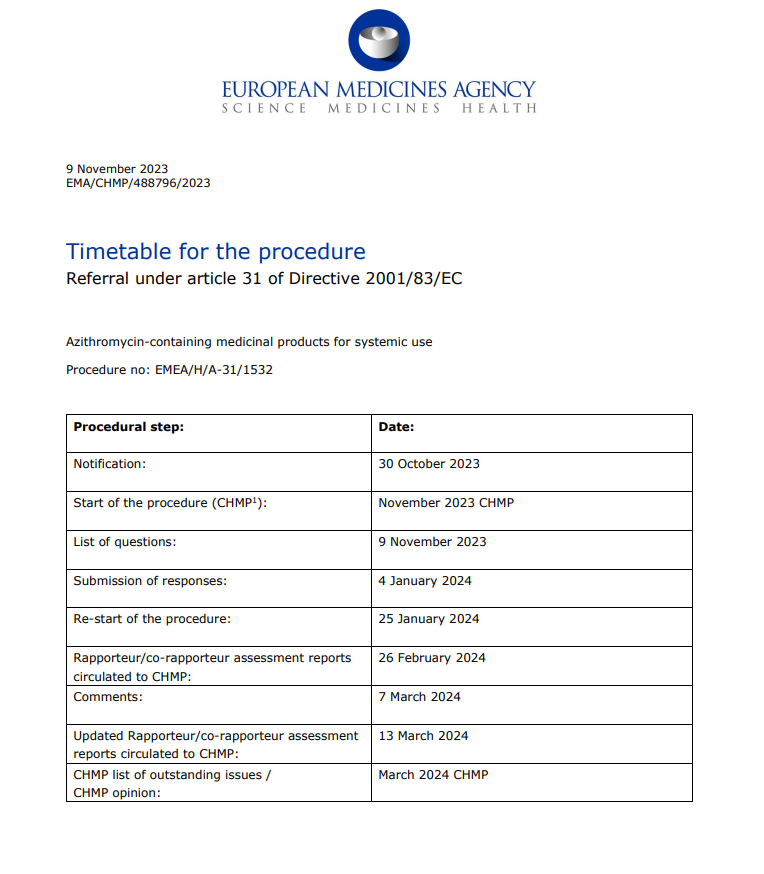
https://ema.europa.eu/en/medicines/human/referrals/azithromycin-containing-medicinal-products-systemic-use uhh germany requested the EMA "review" azithromycin for systemic use.. @jikkyleaks what's going on here?

@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
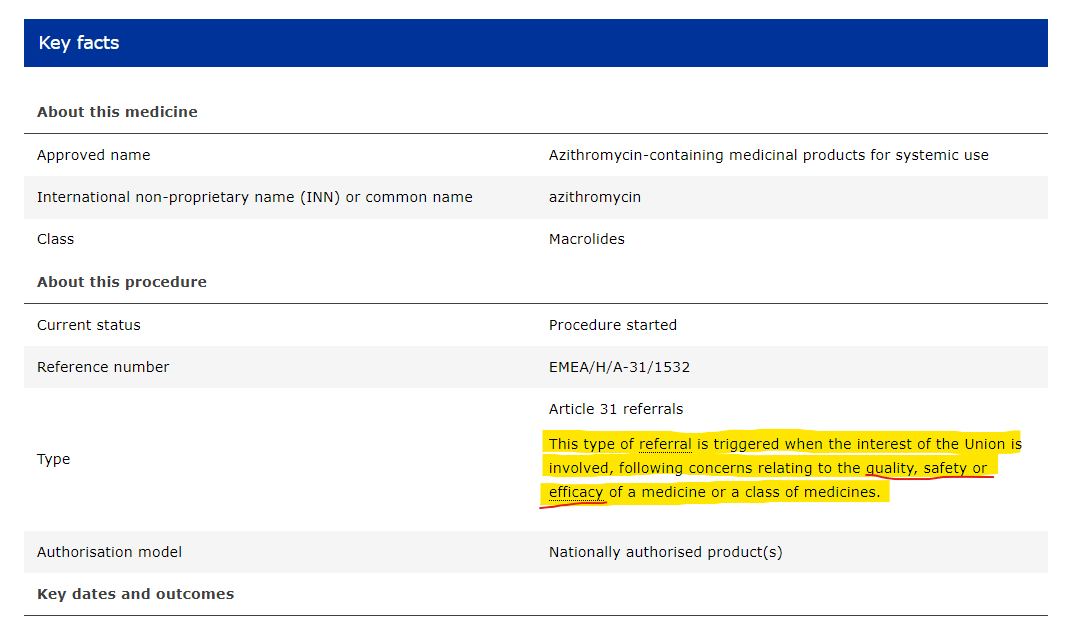
@Jikkyleaks the perversion of truth is undeniable at this point. azithromycin gets a Type 31 referral before the modRNA jabs do. the german government, the EU and EMA must be rebuilt from the foundations up.
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
https://www.ema.europa.eu/en/documents/position/azithromycin-article-31-referral-notification_en.pdf it seems to boil down to "it worked pretty fucking well because it's the first choice and there's no actual evidence of resistance except more bugs in general and more azithro prescribed so we need to review dosing and safety" its outrageous tbh
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
https://www.ema.europa.eu/en/documents/referral/azithromycin-article-31-referral-chmp-list-questions_en.pdf there's a set of eight questions asked of the MAH, no idea who that is for a generic ab? wonder if any of the regs referred to here have been updated lately?

@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
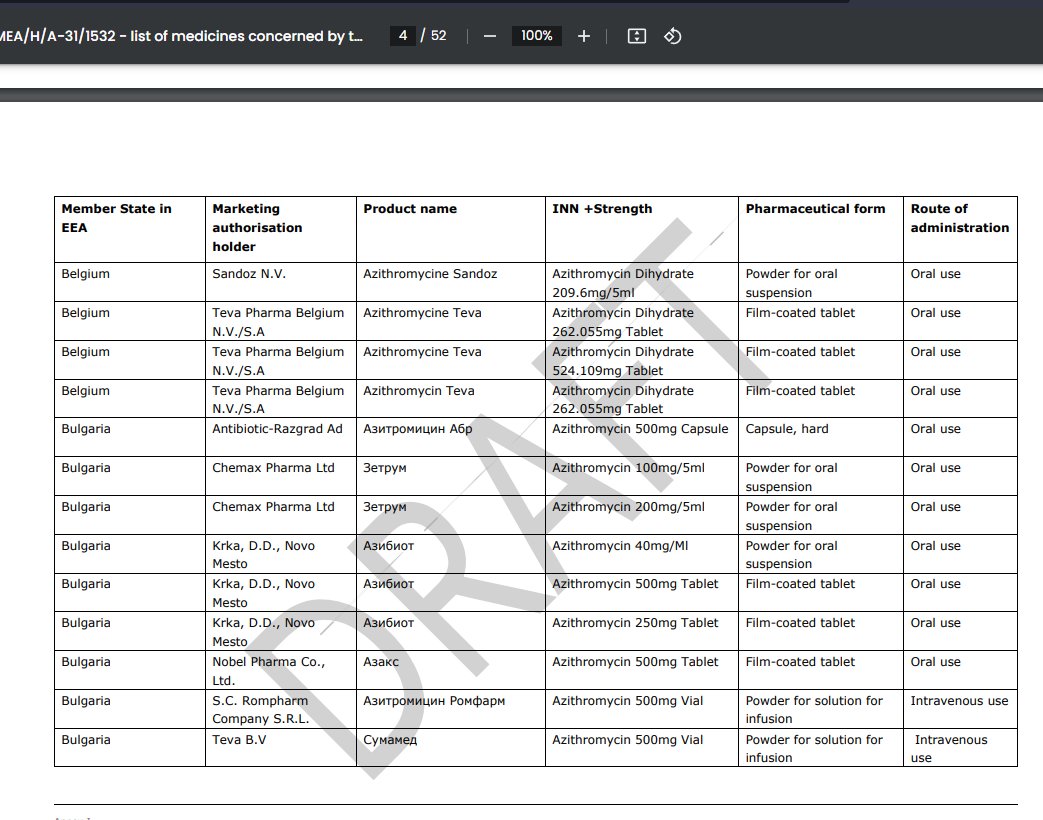
there's also Annex 1, a 52 page list of all forms of AZM marketed across the different member states https://www.ema.europa.eu/en/documents/referral/azithromycin-article-31-referral-annex-i_en.pdf

@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
https://www.ema.europa.eu/documents/referral/azithromycin-article-31-referral-timetable-procedure_en.pdf and finally a timetable for the whole "procedure". the "review" of an actually "safe & effective" antibiotic will be done in four months from now. anybody wanna guess what their decision will be?


@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
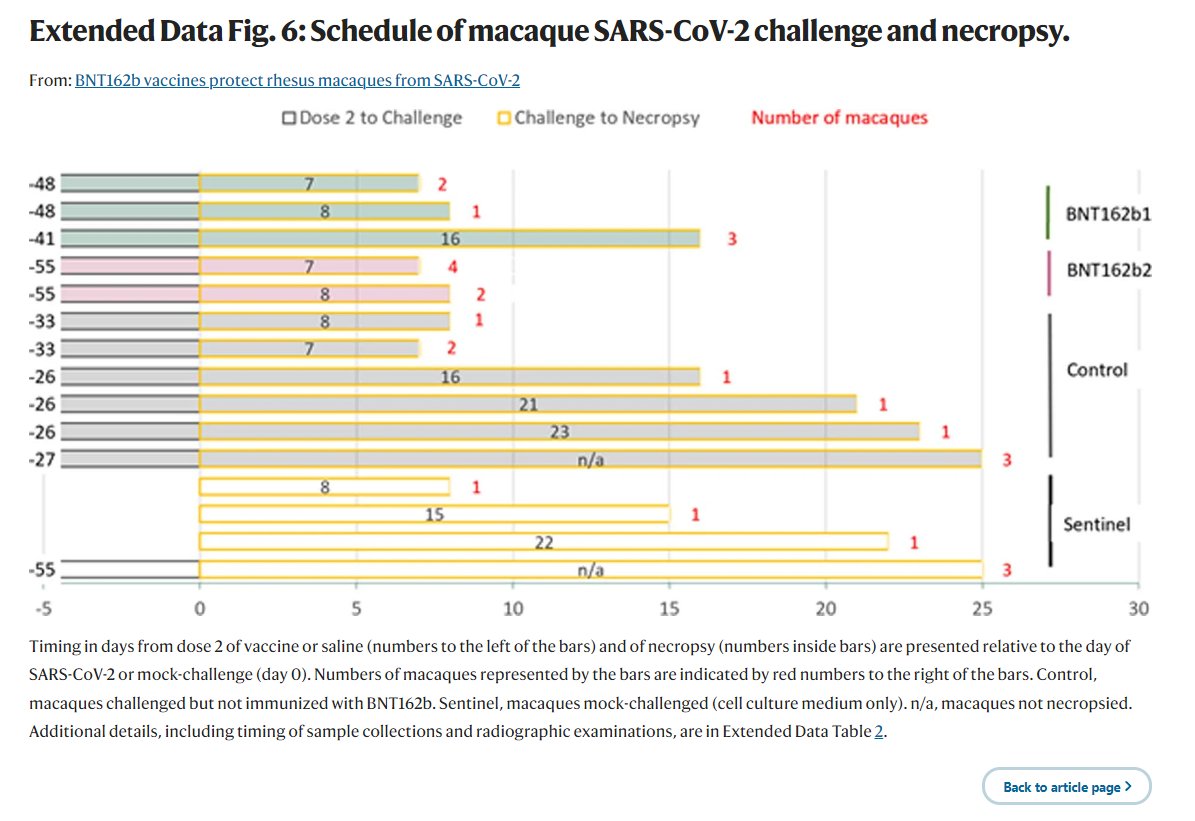
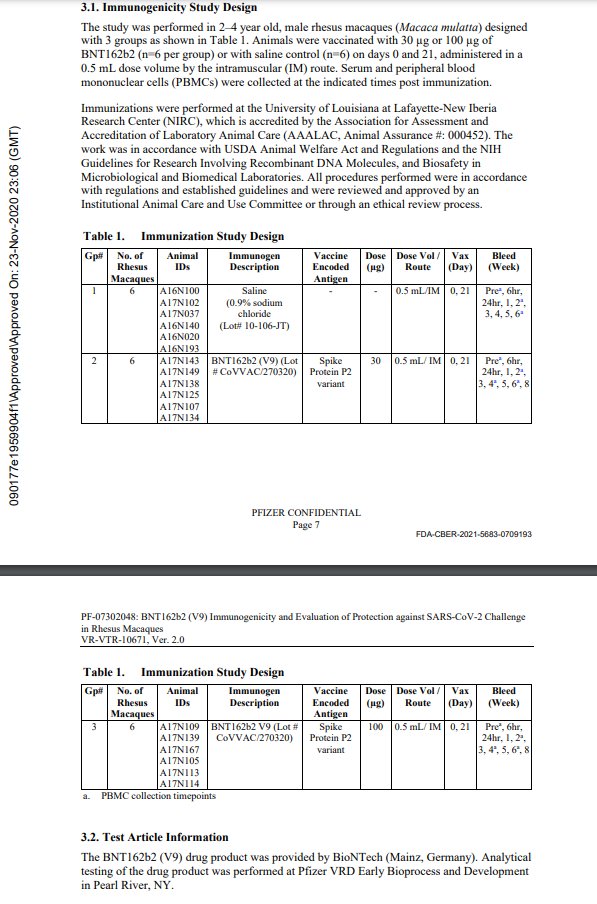
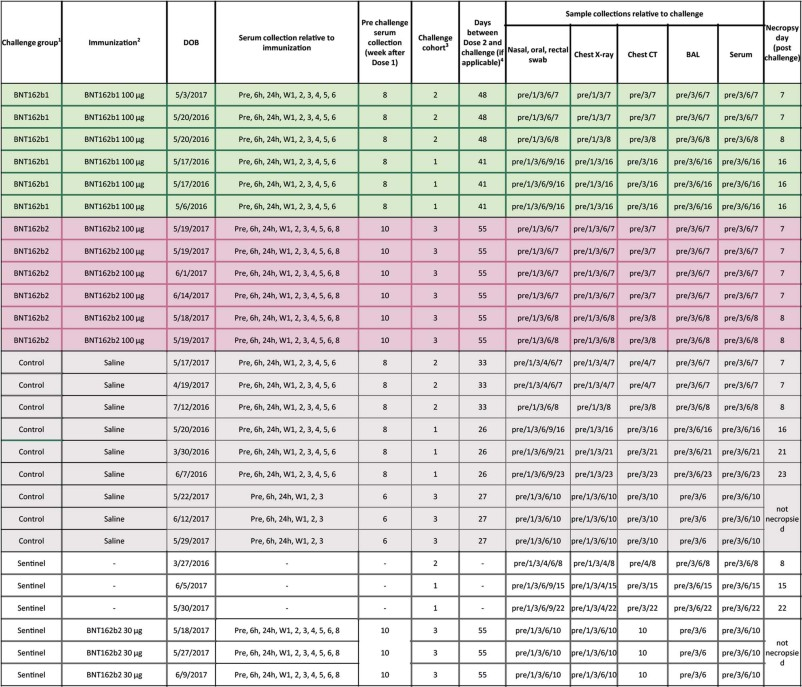
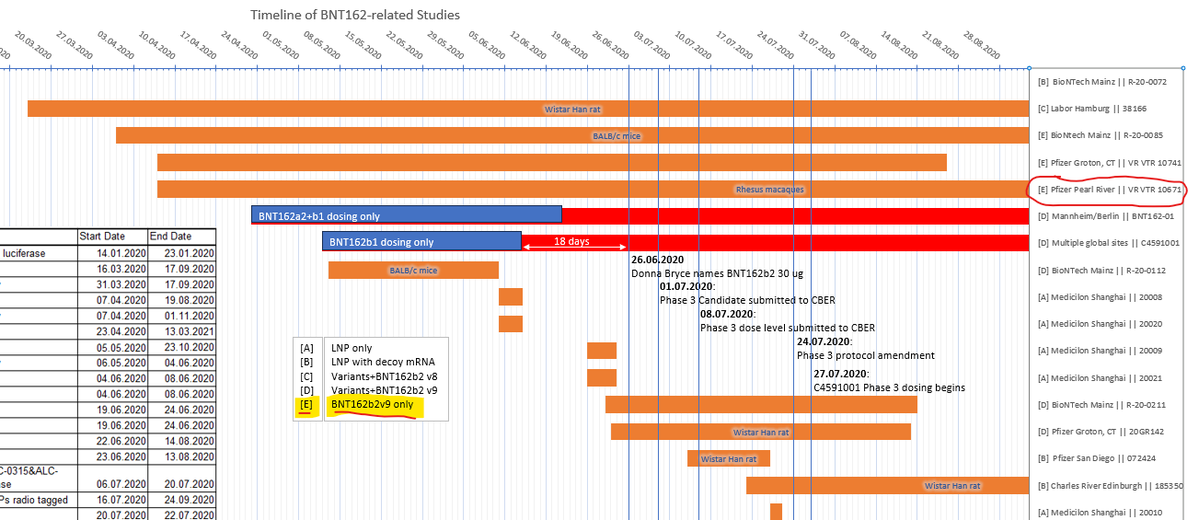
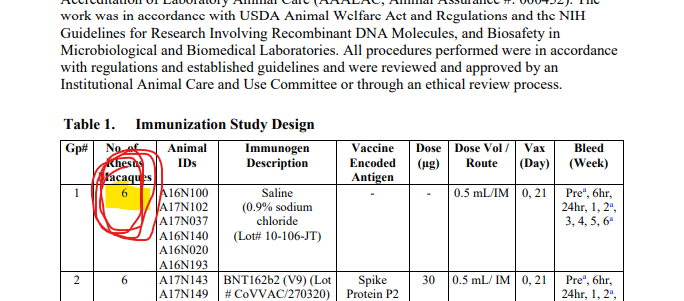
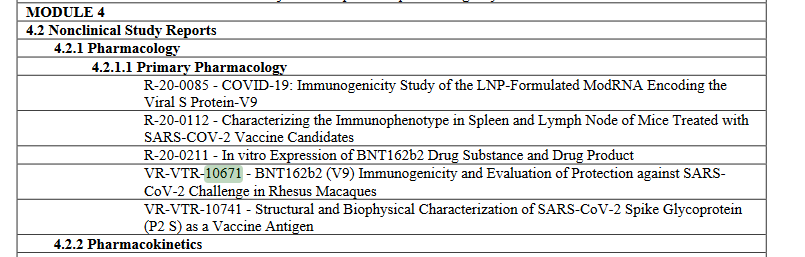
lmao i never realized what pfizer did with the monkey study til now! in the nature paper, they describe using BNT162b1 and BNT162b2 - but PHMPT only has a paper with two dose groups of BNT162b2🤔🤔 https://phmpt.org/wp-content/uploads/2023/02/125742_S1_M4_4.2.2.1-vr-vtr-10671.pdf https://nature.com/articles/s41586-021-03275-y?error=cookies_not_supported&code=a25f5840-ce82-4839-8c29-c9476a513f62…
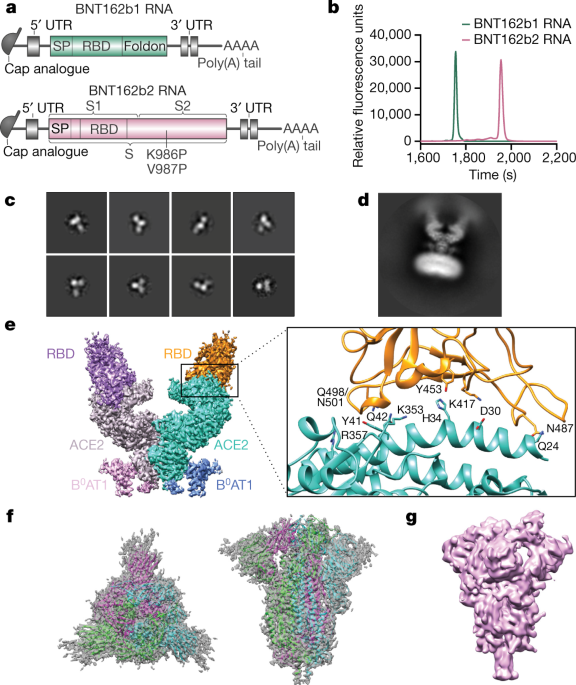
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
the BNT162b1 rhesus macaque experiments have not been published on PHMPT yet. more concerningly, the Nature paper claims 6 b1 & 6 b2-dosed monkeys, while the PHMPT report shows 12 b2-dosed monkeys! the nature paper is missing the 30 ug b2 group 🤔 https://t.co/xxty28vN8t
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
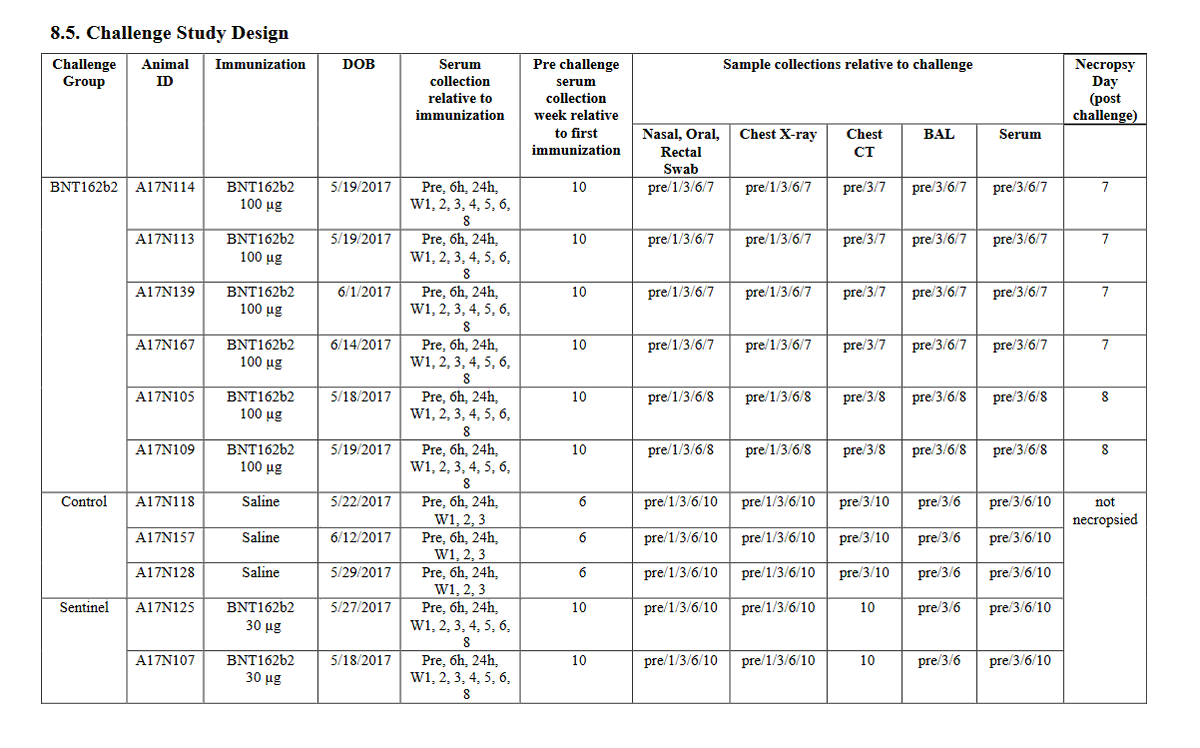
so the submitted pfizer nonclinical study 10671 only includes the data from the Nature paper's "challenge cohort 3" aka. the 100 ug b2-dosed animals that were challenged at the timepoint of maximum vaccine effect🙄 excuse me but wtf? where is the b1 monkey data?
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
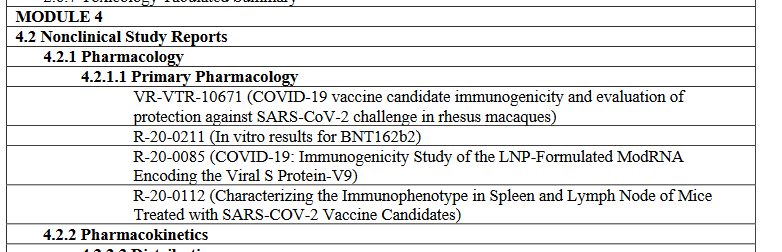
wow it's like the dates are all made up and don't matter at all. this is now the third nonclinical study i've found claiming to have initiated experiments BEFORE the corresponding nonclin batch was released by polymun(38166 and R-20-0085 also have this "problem") https://t.co/NPIvccUiuv
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
it also puts me in a bit of a pickle because do i mark 10671 as b2v9 only [E] or b2v9+variants [D]? did pfizer also submit the b1 data in the BLA or only in the Nature paper? reading between the lines it sure makes b2 seem like the actual candidate all along. recurring theme.. https://t.co/uUV31VxHtm
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
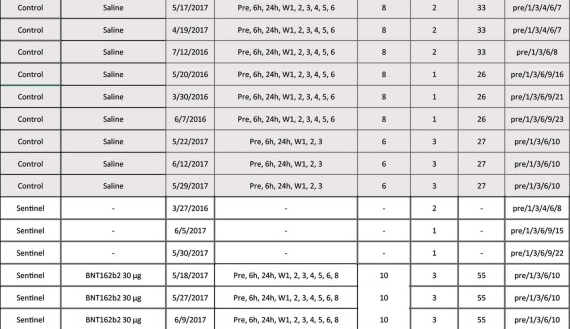
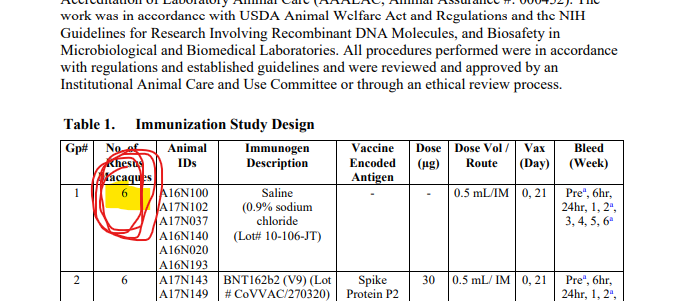
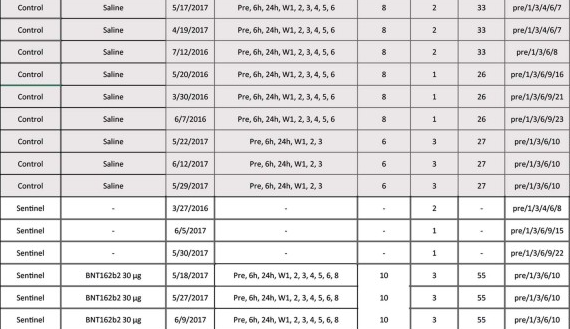
LOL, 10671 on PHMPT has 6 control monkeys, Nature paper has NINE and six vehicle, the latter not even being included in the PHMPT count. smooth operators all around! nothing like a bit of cherrypicking to take the edge off https://t.co/1Mzjuf96Fq
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
first screenie is p477, next is p543 of meeting-correspondence https://phmpt.org/wp-content/uploads/2023/10/125742_S1_M1_meeting-correspondence.pdf. notice how the study description changed in between: "vaccine candidates" -> BNT162b2v9 only. the nature paper was submitted september 1st 2020 🤔🤔 https://t.co/iuBxunJjZM
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
in other words, the first version on 10671 the nature paper is based on wasnt the version of sthe study report that was submitted to the EUA/BLA. they took out the b1 data and added another b2 dosing cohort. why, and more importantly, how? why didnt regulators call this out??
@a_nineties - a_concerned_amyloidosis💜🐭🇩🇪
haha it's even worse than i thought, they cherrypicked 3 controls and three vehicle monkeys and omitted all control necropsy data from the PHMPT report 😂https://t.co/0rpq98UCdO https://t.co/NssDO9eK1R