TruthArchive.ai - Tweets Saved By @joshg99

@joshg99 - Josh Guetzkow
What?!? In 2022 & 2023, the FDA cited significant violations at an EU facility where Pfizer's mRNA 'drug substance' is purified prior to LNP encapsulation. Violations including failure to assure compliance w/specifications & standards for endotoxin testing. But there's more! 🧵
@joshg99 - Josh Guetzkow
Background: Pfizer used a new process scale up production for the commercial product (different from the one it tested). Using plasmids grown in E.coli bacteria, the new process introduced DNA & endotoxin contamination that required thorough purification. https://x.com/joshg99/status/1658421192326365185
@joshg99 - Josh Guetzkow
In Oct. 2020, Rentschler Biopharma announced its contract w/Pfizer for "downstream processing to provide highly purified drug substance" where process-related "impurities will be effectively removed" at its Laupheim, Baden-Württemberg facility in Germany. https://www.rentschler-biopharma.com/news/press-releases-and-announcements/detail/view/joining-forces-against-sars-cov-2

@joshg99 - Josh Guetzkow
In Feb '22 the FDA notified the facility of 9 violations including the endotoxin testing problem above, their samples for testing were not representative of the final product & no system to alert them to ultracold storage freezer failures they found. https://www.fiercepharma.com/manufacturing/rentschler-slapped-form-483-citing-lax-manufacturing-procedures

@joshg99 - Josh Guetzkow
In March '23 the FDA again notified the facility of 5 new violations, including poor monitoring of endotoxin contamination of equipment and other procedures inadequate to prevent microbial contamination. https://www.fiercepharma.com/manufacturing/manufacturing-operations-eli-lilly-rentschler-reprimanded-fda-form-483-filings

@joshg99 - Josh Guetzkow
Why does this matter? In 2021 Pfizer started a study to compare multiple production lots of its COVID vaccine for safety, tolerability, and immunogenicity. It seems they were all lots made with process 2, which was used for commercial supply.
@joshg99 - Josh Guetzkow
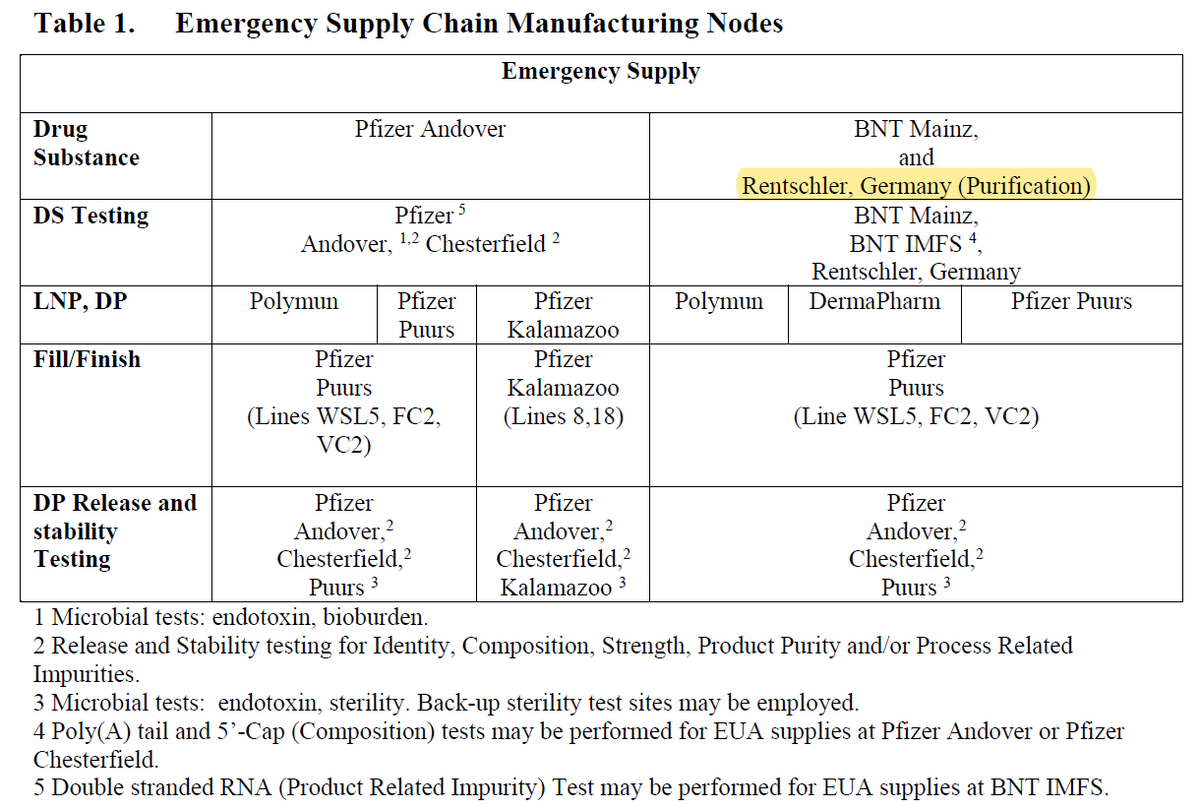
The study compared 3 lots made in the US to 1 lot made in the EU. This table from a leaked FDA document indicates that all purification of drug substance made in Europe was done at the Rentschler site.
@joshg99 - Josh Guetzkow
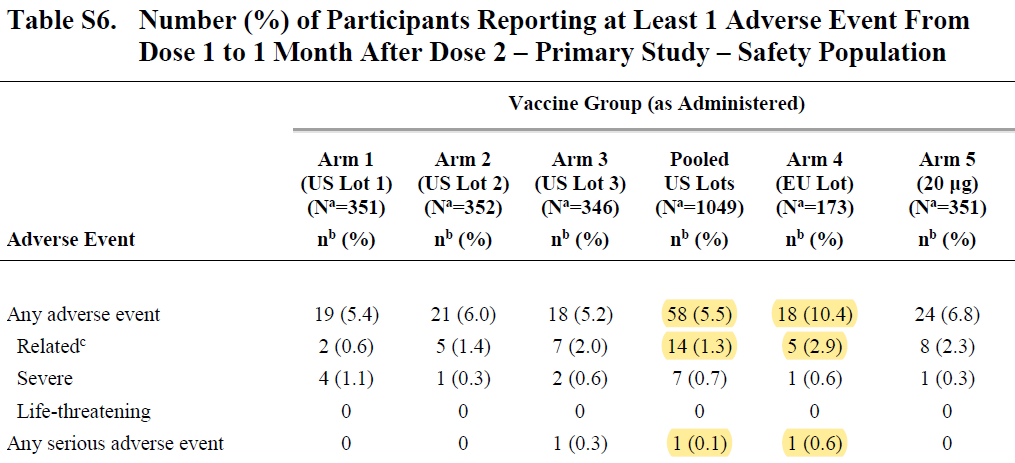
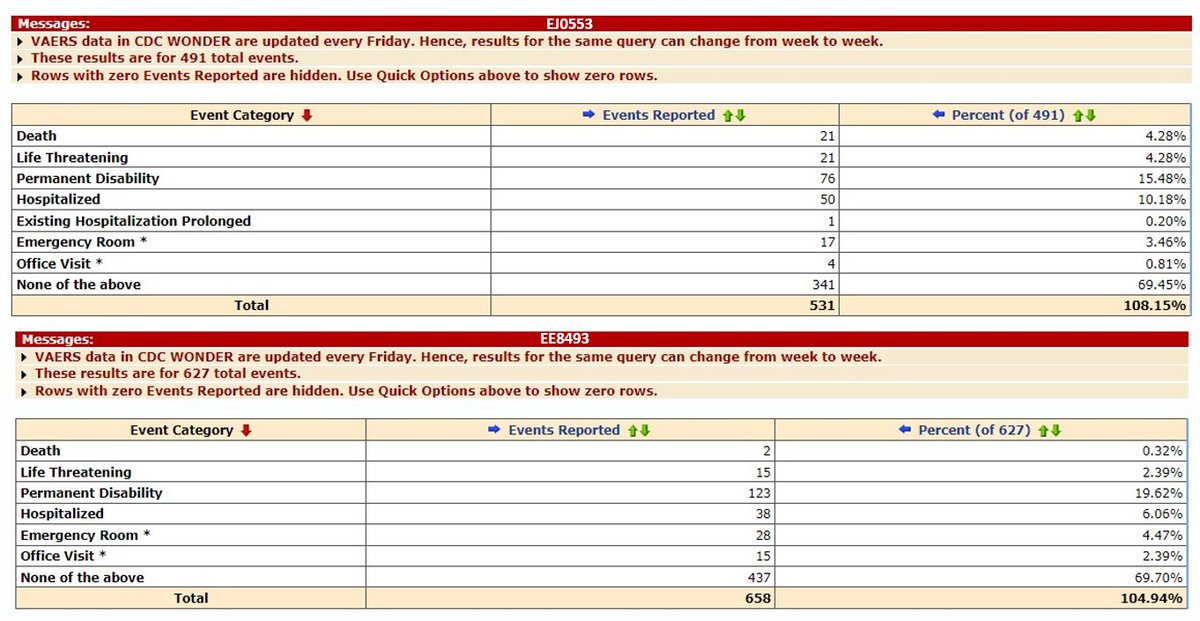
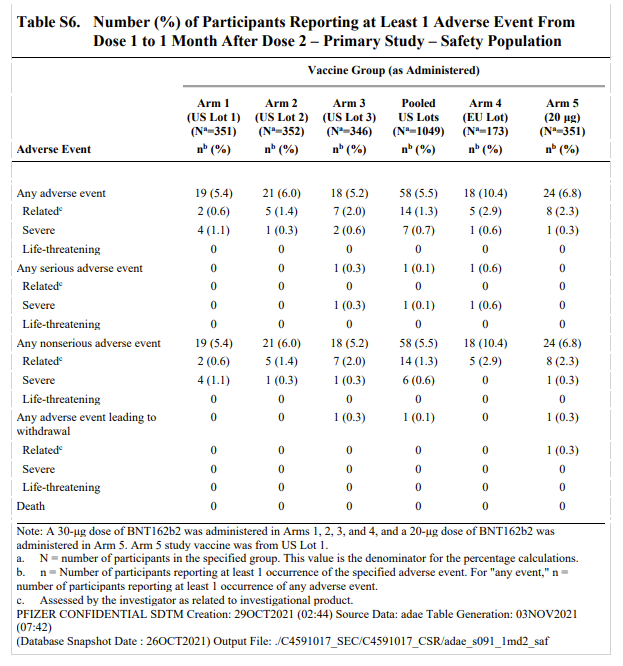
The comparability study found the EU lot had double the rate of adverse events as the combined US lots -- a difference which is statistically significant. The EU lot had 1 serious AE out of 173 subjects compared with 1 SAE from US lots with 1,049 participants (0.1% vs. 0.6%).
@joshg99 - Josh Guetzkow
In short, Pfizer's own lot-to-lot comparison study shows a significant difference in the safety of doses made in the US vs. doses made in the EU that passed through this manufacturing plant in Germany for purification prior to being encapsulated in LNPs at a different site.
@joshg99 - Josh Guetzkow
The tests Pfizer used to check for DNA and endotoxin are not suited to detect contamination encapsulated in the LNPs. That's why purification prior to encapsulation is so important. And may explain at least in part the increased dangers of the EU-manufactured lots.
@joshg99 - Josh Guetzkow
For more on the dangers of DNA contamination in the mRNA vaccines, see this pre-print: https://osf.io/mjc97/ For more on the dangers of endotoxin contamination see Geoff Pain's blog (endotoxins are pieces of E.coli membranes): https://geoffpain.substack.com/


@joshg99 - Josh Guetzkow
Big thanks to @Quo_vadis_BRD for bringing the FDA reports to my attention & @a_nineties for clarifying important issues related to Pfizer's comparability study.

@joshg99 - Josh Guetzkow
Was the Pfizer/BioNTech vaccine clinical trial a bait-and-switch? There were >44,000 people in the trial, but only ~250 of them were given doses made with a new manufacturing method ('process 2') that was used to make enough doses to sell around the world.
@joshg99 - Josh Guetzkow
To our knowledge, the safety and efficacy comparison they planned to do with those 250 subjects has never been published and has not been released in the FOIA'd documents that Pfizer submitted to the FDA. Was the comparison ever done? Where are the results?
@joshg99 - Josh Guetzkow
@RetsefL and I explore the importance of this comparison and the potential impact of variability in the the production process of COVID-19 mRNA vaccines on efficacy and safety in a newly published rapid response in the @bmj_latest. https://www.bmj.com/content/378/bmj.o1731/rr-2

@joshg99 - Josh Guetzkow
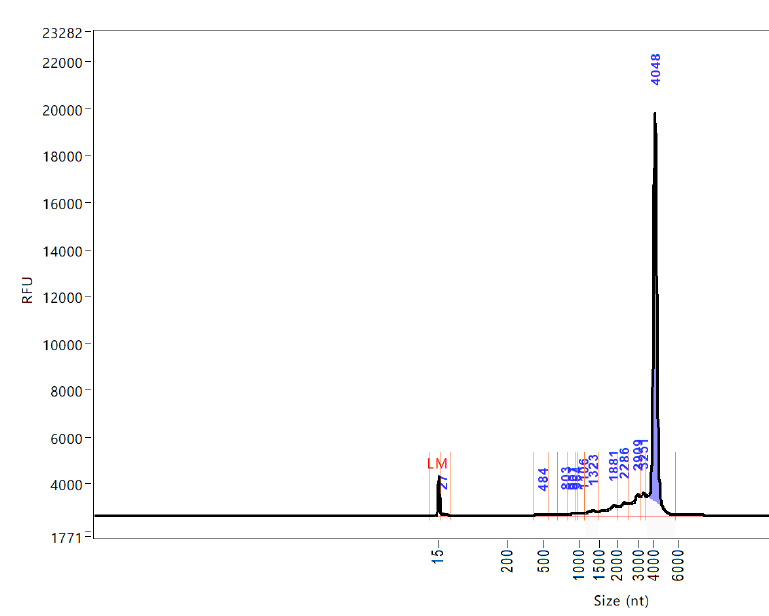
Keep in mind that one of the major changes in the new production process was using bacterial cDNA to upscale production of mRNA. @Kevin_McKernan's analysis of vaccine vials found unacceptably high levels of leftover bacterial DNA. https://www.rebelnews.com/genomics_expert_discovers_concerning_contents_in_covid_vaccine_vials

@joshg99 - Josh Guetzkow
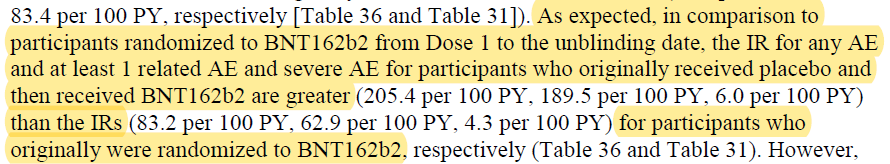
Pfizer's 6-month report to the FDA doesn't include the process 2 comparison, but it does show a significantly higher serious adverse event rate in placebo subjects after they were given the vaccine compared to the original vaccine group, "as expected." Why was it expected?
@joshg99 - Josh Guetzkow
We know from FOIA'd documents that about 70% of the trial sites received new batches with distinct Pfizer lot numbers after Nov. 19. Were these intended for the crossover placebo subjects? Were they different than the doses given to the original treatment group? And if so, how?
@joshg99 - Josh Guetzkow
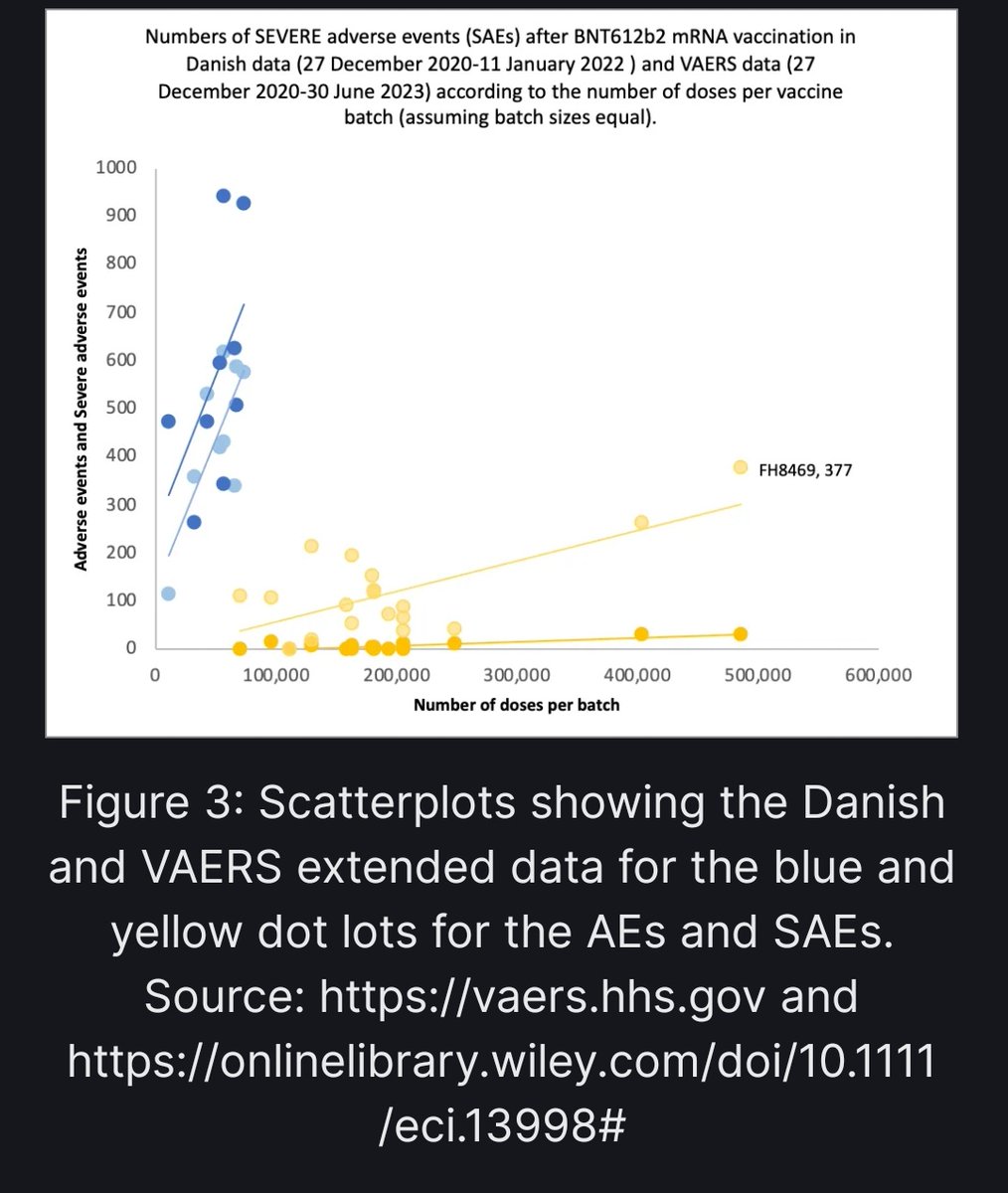
In addition, a recent Danish study found significant variability in the rate of serious adverse events across 52 different lots of Comirnaty. https://onlinelibrary.wiley.com/doi/10.1111/eci.13998
@joshg99 - Josh Guetzkow
Taken together, evidence from trial documents and existing research underscores the need to better understand the potential impact of variability in the production process of COVID-19 mRNA vaccines on efficacy and safety.
@joshg99 - Josh Guetzkow
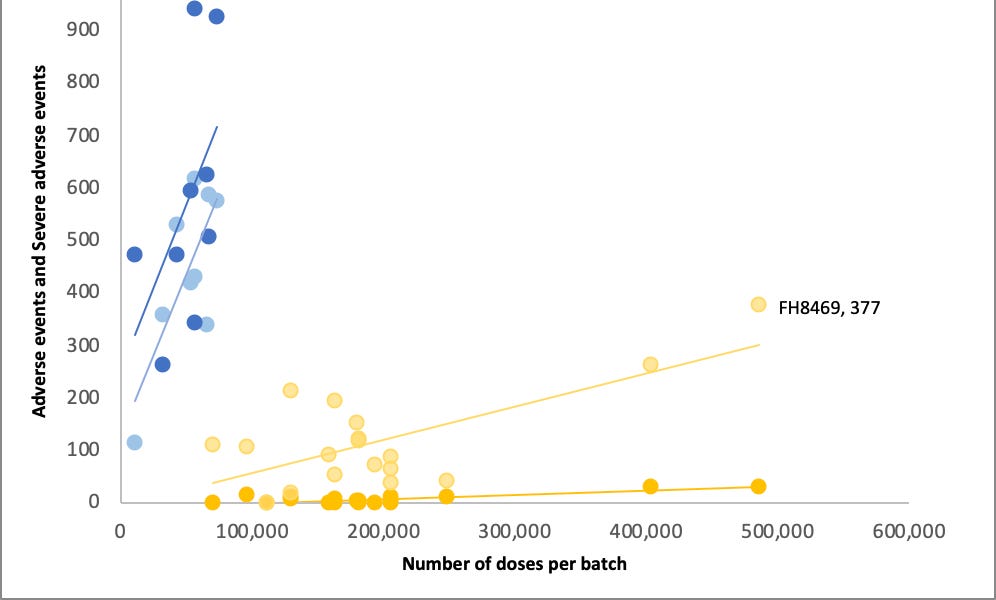
I forgot to add: The two process 2 lots sent to 8 trial sites were also given to the public. There are 1,149 reports in VAERS for these lots, including 307 serious adverse events and 23 deaths.
@joshg99 - Josh Guetzkow
This thread is also highly relevant to manufacturing standards and batch toxicity:
@joshg99 - Josh Guetzkow
Somebody asked if study C4591017 was the process 2 comparison trial: https://clinicaltrialsregister.eu/ctr-search/trial/2021-005903-11/results It isn't though it does compare 5 (unidentified) lots & found a 2x rate of subjects experiencing at least one AE and AEs judged related to the vaccine in one lot & variation in SAEs.
@joshg99 - Josh Guetzkow
In case it isn't clear, the 2 production processes were radically different and yielded different quality products! You can't just test it on 250 people and say it's all the same. They could not be assumed to be bio-similar, like name-brand and generic.
@joshg99 - Josh Guetzkow
This article has excellent coverage of this issue. It also points out the fact that the manufacturer was never required to test process 2 jabs on lab animals. Or at least we've never seen results of such tests. https://www.conservativewoman.co.uk/how-hancock-and-the-fiercely-independent-mhra-helped-pfizer-game-the-vaccine-approval-process/

@joshg99 - Josh Guetzkow
Genomics expert, professor @P_J_Buckhaults U of South Carolina, testifies on the potential risks of the DNA contamination he found in vials of mRNA vaccine entirely that was entirely due to the switch to process 2. https://x.com/TheChiefNerd/status/1703544942004478251
@joshg99 - Josh Guetzkow
However I suspect that the endotoxin contamination is likely a bigger problem, both because it is probably a major factor in many adverse events and because it is basically impossible to purify. For more on endotoxin risks: https://geoffpain.substack.com/

@joshg99 - Josh Guetzkow
See this thread for a further update showing that Pfizer/BioNTech never did the planned study comparing process 2 v 1 for safety and immunogenicity: https://x.com/joshg99/status/1705177305499279525
@joshg99 - Josh Guetzkow
A professor and expert in cancer genetics at U of South Carolina testifies on risks posed by process 2 jabs due to the plasmid DNA contamination: https://www.youtube.com/watch?v=IEWHhrHiiTY
@joshg99 - Josh Guetzkow
And a transcript of his interview with Maryanne Demasi: https://maryannedemasi.substack.com/p/exclusive-an-interview-with-buckhaults
@joshg99 - Josh Guetzkow
Great talk by @hedleyrees discussing the intricacies and challenges of biologic drug manufacturing: https://rumble.com/v3fqujk-challenges-in-manufacturing-and-distributing-products-derived-from-biologic.html


@joshg99 - Josh Guetzkow
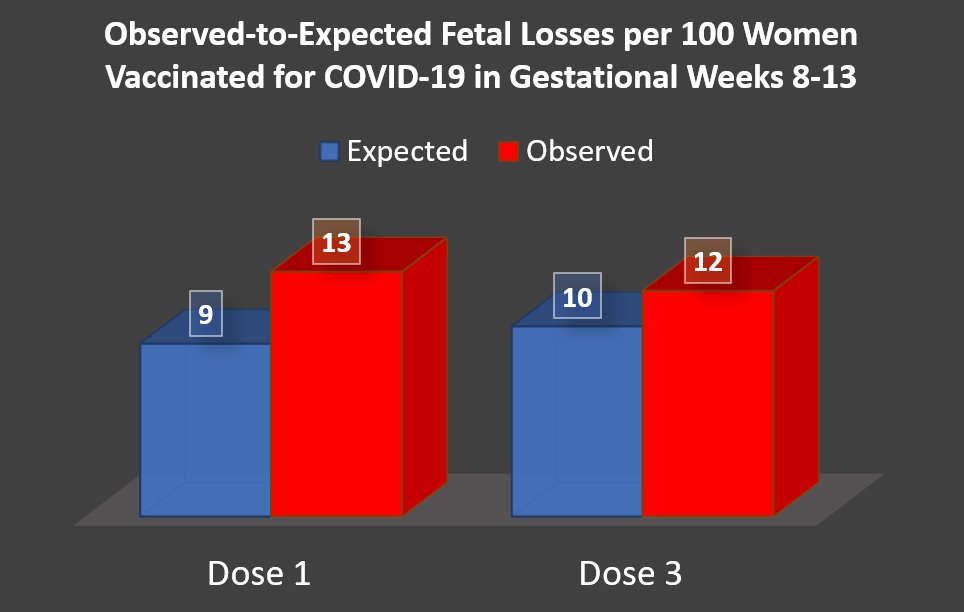
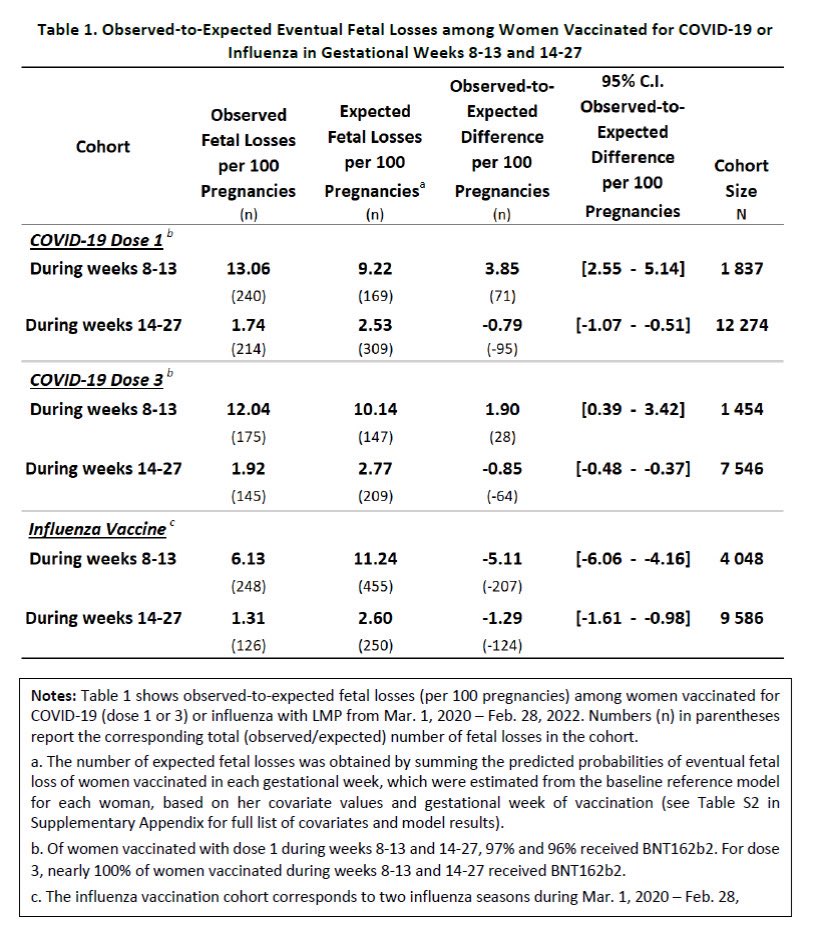
Our new pre-print on COVID-19 vaccine safety in pregnancy is out! Women vaccinated in early pregnancy (weeks 8-13) had a higher-than-expected number of fetal losses: Dose 1 = 3.9 more per 100 women Dose 3 = 1.9 more per 100 women Based on data on >200K pregnancies in Israel 🧵
@joshg99 - Josh Guetzkow
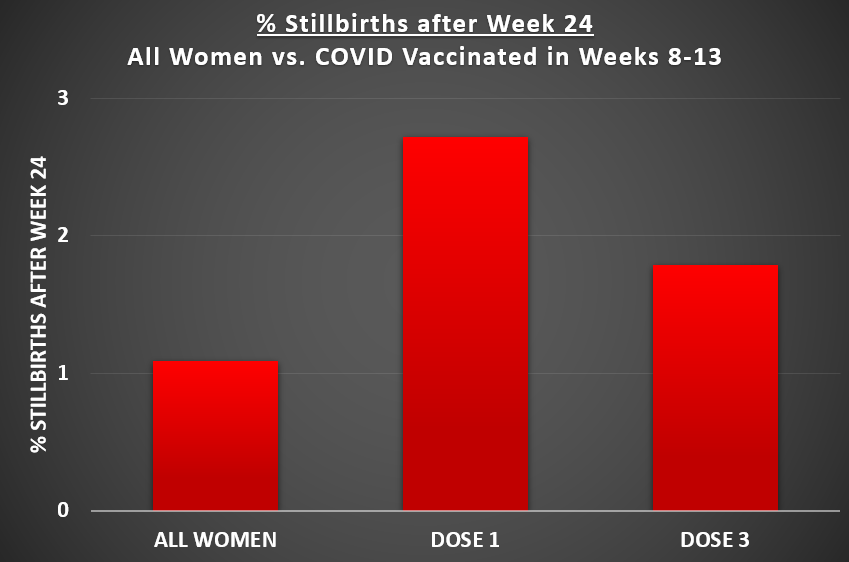
2/ Late pregnancy losses were a *big* part of the signal. Among all women: 1.1% lost their pregnancy after week 24. Compared to: 2.7% of women who received dose 1 in early weeks. 1.8% of women who received dose 3 in early weeks.
@joshg99 - Josh Guetzkow
3/ In fact, most of the excess losses occurred later in pregnancy, including nearly half after week 24. In Israel, abortions after week 24 are rare and must be medically justified and are very rare. This strongly suggests biological, not behavioral, mechanisms are involved.
@joshg99 - Josh Guetzkow
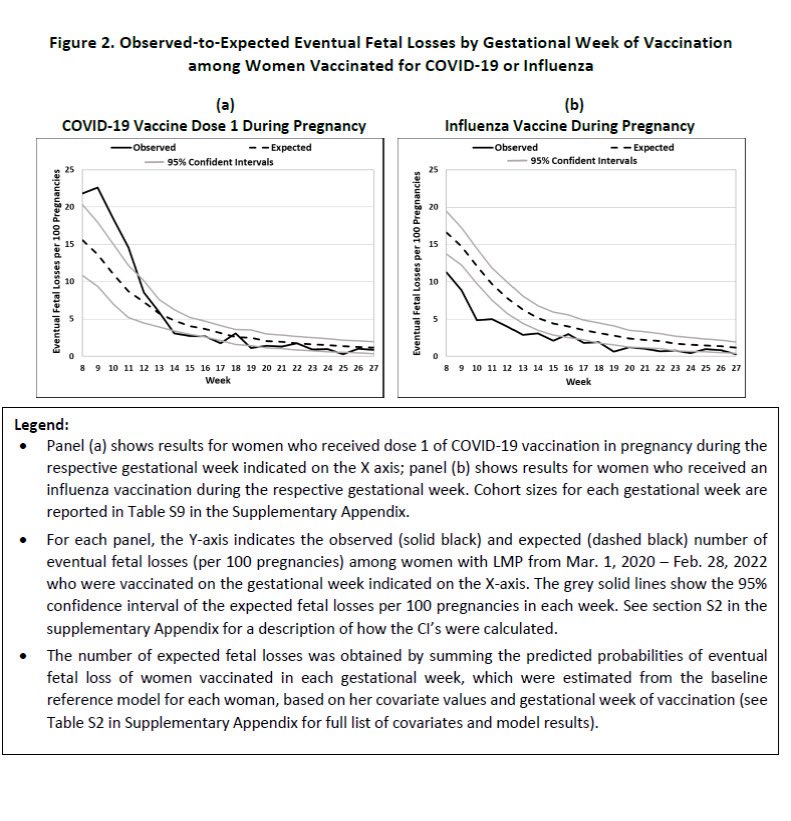
4/ In stark contrast, women vaccinated for influenza in the same weeks (8-13) saw *FEWER* fetal losses than expected, about 5 per 100 women vaccinated. What's the significance of that?
@joshg99 - Josh Guetzkow
5/ Comparing both types of vaccines helps control for bias in who chooses to get vaccinated. For example, if women vaccinate in early pregnancy because they have health problems, the elevated fetal loss risk for COVID-19 vaccines could simply be due to that bias.
@joshg99 - Josh Guetzkow
6/ But people who get vaccinated are usually healthier & more health conscious to begin with. This is called healthy vaccinee bias & can make vaccines look safer than they really are. It's a known – but often ignored -- issue in vaccine safety research based on real-world data.
@joshg99 - Josh Guetzkow
7/ Influenza vaccination during pregnancy showed lower-than-expected fetal losses—likely due to healthy vaccine bias. So it's even more striking that mRNA COVID-19 vaccination in early pregnancy showed higher-than-expected fetal losses—despite healthy vaccinee bias.
@joshg99 - Josh Guetzkow
8/ Our findings don’t prove causation. But they point to a clear safety signal—one that calls for further investigation, especially given the vulnerability of this population.
@joshg99 - Josh Guetzkow
9/ So why hasn’t this been seen before? Most other studies compare vaccinated vs. unvaccinated women during vaccination campaigns. That approach is vulnerable to bias—especially if healthier women are more likely to vaccinate (a.k.a. “healthy vaccinee bias”).
@joshg99 - Josh Guetzkow
10/ Our approach was different. We used detailed medical records to estimate expected fetal loss rates for each vaccinated woman—based on her individual risk factors and pregnancy timing—using data from pre-COVID years.
@joshg99 - Josh Guetzkow
11/ Here is a link to the pre-print: https://medrxiv.org/cgi/content/short/2025.06.18.25329352v1 This was of course a team effort with my amazing co-authors: @RetsefL, @TalPatalon, Sivan Gazit, @TracyBethHoeg, @JosephFraiman and Yaakov Segal.

@joshg99 - Josh Guetzkow

@joshg99 - Josh Guetzkow
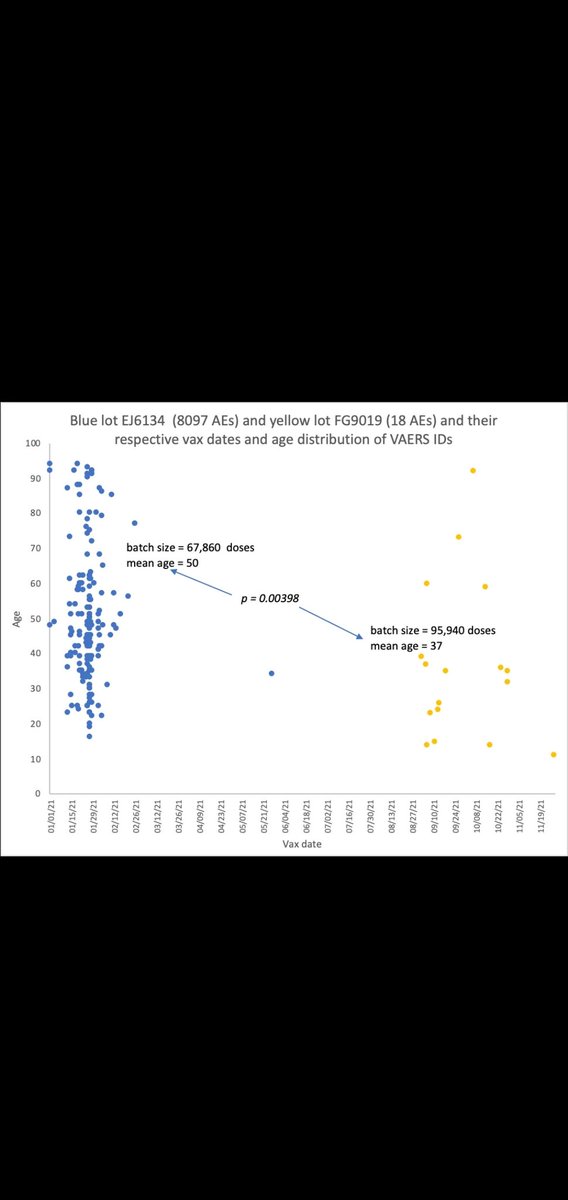
@JesslovesMJK nails the coffin on the placebo theory. Great work! Age and date profile is totally different and SAEs abound. Yellow dots prob. used for boosters?@ChrisEd16512812@FluoridePoison@EdV1694@TheChiefNerd @denisrancourt https://open.substack.com/pub/jessicar/p/debunking-the-yellow-dot-lot-placebo

@joshg99 - Josh Guetzkow
What?!? In 2022 & 2023, the FDA cited significant violations at an EU facility where Pfizer's mRNA 'drug substance' is purified prior to LNP encapsulation. Violations including failure to assure compliance w/specifications & standards for endotoxin testing. But there's more! 🧵
@joshg99 - Josh Guetzkow
Background: Pfizer used a new process scale up production for the commercial product (different from the one it tested). Using plasmids grown in E.coli bacteria, the new process introduced DNA & endotoxin contamination that required thorough purification. https://x.com/joshg99/status/1658421192326365185
@joshg99 - Josh Guetzkow
In Oct. 2020, Rentschler Biopharma announced its contract w/Pfizer for "downstream processing to provide highly purified drug substance" where process-related "impurities will be effectively removed" at its Laupheim, Baden-Württemberg facility in Germany. https://www.rentschler-biopharma.com/news/press-releases-and-announcements/detail/view/joining-forces-against-sars-cov-2

@joshg99 - Josh Guetzkow
In Feb '22 the FDA notified the facility of 9 violations including the endotoxin testing problem above, their samples for testing were not representative of the final product & no system to alert them to ultracold storage freezer failures they found. https://www.fiercepharma.com/manufacturing/rentschler-slapped-form-483-citing-lax-manufacturing-procedures

@joshg99 - Josh Guetzkow
In March '23 the FDA again notified the facility of 5 new violations, including poor monitoring of endotoxin contamination of equipment and other procedures inadequate to prevent microbial contamination. https://www.fiercepharma.com/manufacturing/manufacturing-operations-eli-lilly-rentschler-reprimanded-fda-form-483-filings

@joshg99 - Josh Guetzkow
Why does this matter? In 2021 Pfizer started a study to compare multiple production lots of its COVID vaccine for safety, tolerability, and immunogenicity. It seems they were all lots made with process 2, which was used for commercial supply.
@joshg99 - Josh Guetzkow
The study compared 3 lots made in the US to 1 lot made in the EU. This table from a leaked FDA document indicates that all purification of drug substance made in Europe was done at the Rentschler site.
@joshg99 - Josh Guetzkow
The comparability study found the EU lot had double the rate of adverse events as the combined US lots -- a difference which is statistically significant. The EU lot had 1 serious AE out of 173 subjects compared with 1 SAE from US lots with 1,049 participants (0.1% vs. 0.6%).
@joshg99 - Josh Guetzkow
In short, Pfizer's own lot-to-lot comparison study shows a significant difference in the safety of doses made in the US vs. doses made in the EU that passed through this manufacturing plant in Germany for purification prior to being encapsulated in LNPs at a different site.
@joshg99 - Josh Guetzkow
The tests Pfizer used to check for DNA and endotoxin are not suited to detect contamination encapsulated in the LNPs. That's why purification prior to encapsulation is so important. And may explain at least in part the increased dangers of the EU-manufactured lots.
@joshg99 - Josh Guetzkow
For more on the dangers of DNA contamination in the mRNA vaccines, see this pre-print: https://osf.io/mjc97/ For more on the dangers of endotoxin contamination see Geoff Pain's blog (endotoxins are pieces of E.coli membranes): https://geoffpain.substack.com/


@joshg99 - Josh Guetzkow
Big thanks to @Quo_vadis_BRD for bringing the FDA reports to my attention & @a_nineties for clarifying important issues related to Pfizer's comparability study.

@joshg99 - Josh Guetzkow
Was the Pfizer/BioNTech vaccine clinical trial a bait-and-switch? There were >44,000 people in the trial, but only ~250 of them were given doses made with a new manufacturing method ('process 2') that was used to make enough doses to sell around the world.
@joshg99 - Josh Guetzkow
To our knowledge, the safety and efficacy comparison they planned to do with those 250 subjects has never been published and has not been released in the FOIA'd documents that Pfizer submitted to the FDA. Was the comparison ever done? Where are the results?
@joshg99 - Josh Guetzkow
@RetsefL and I explore the importance of this comparison and the potential impact of variability in the the production process of COVID-19 mRNA vaccines on efficacy and safety in a newly published rapid response in the @bmj_latest. https://www.bmj.com/content/378/bmj.o1731/rr-2

@joshg99 - Josh Guetzkow
Keep in mind that one of the major changes in the new production process was using bacterial cDNA to upscale production of mRNA. @Kevin_McKernan's analysis of vaccine vials found unacceptably high levels of leftover bacterial DNA. https://www.rebelnews.com/genomics_expert_discovers_concerning_contents_in_covid_vaccine_vials

@joshg99 - Josh Guetzkow
Pfizer's 6-month report to the FDA doesn't include the process 2 comparison, but it does show a significantly higher serious adverse event rate in placebo subjects after they were given the vaccine compared to the original vaccine group, "as expected." Why was it expected?
@joshg99 - Josh Guetzkow
We know from FOIA'd documents that about 70% of the trial sites received new batches with distinct Pfizer lot numbers after Nov. 19. Were these intended for the crossover placebo subjects? Were they different than the doses given to the original treatment group? And if so, how?
@joshg99 - Josh Guetzkow
In addition, a recent Danish study found significant variability in the rate of serious adverse events across 52 different lots of Comirnaty. https://onlinelibrary.wiley.com/doi/10.1111/eci.13998
@joshg99 - Josh Guetzkow
Taken together, evidence from trial documents and existing research underscores the need to better understand the potential impact of variability in the production process of COVID-19 mRNA vaccines on efficacy and safety.
@joshg99 - Josh Guetzkow
I forgot to add: The two process 2 lots sent to 8 trial sites were also given to the public. There are 1,149 reports in VAERS for these lots, including 307 serious adverse events and 23 deaths.
@joshg99 - Josh Guetzkow
This thread is also highly relevant to manufacturing standards and batch toxicity:
@joshg99 - Josh Guetzkow
Somebody asked if study C4591017 was the process 2 comparison trial: https://clinicaltrialsregister.eu/ctr-search/trial/2021-005903-11/results It isn't though it does compare 5 (unidentified) lots & found a 2x rate of subjects experiencing at least one AE and AEs judged related to the vaccine in one lot & variation in SAEs.
@joshg99 - Josh Guetzkow
In case it isn't clear, the 2 production processes were radically different and yielded different quality products! You can't just test it on 250 people and say it's all the same. They could not be assumed to be bio-similar, like name-brand and generic.
@joshg99 - Josh Guetzkow
This article has excellent coverage of this issue. It also points out the fact that the manufacturer was never required to test process 2 jabs on lab animals. Or at least we've never seen results of such tests. https://www.conservativewoman.co.uk/how-hancock-and-the-fiercely-independent-mhra-helped-pfizer-game-the-vaccine-approval-process/

@joshg99 - Josh Guetzkow
Genomics expert, professor @P_J_Buckhaults U of South Carolina, testifies on the potential risks of the DNA contamination he found in vials of mRNA vaccine entirely that was entirely due to the switch to process 2. https://x.com/TheChiefNerd/status/1703544942004478251
@joshg99 - Josh Guetzkow
However I suspect that the endotoxin contamination is likely a bigger problem, both because it is probably a major factor in many adverse events and because it is basically impossible to purify. For more on endotoxin risks: https://geoffpain.substack.com/

@joshg99 - Josh Guetzkow
See this thread for a further update showing that Pfizer/BioNTech never did the planned study comparing process 2 v 1 for safety and immunogenicity: https://x.com/joshg99/status/1705177305499279525
@joshg99 - Josh Guetzkow
A professor and expert in cancer genetics at U of South Carolina testifies on risks posed by process 2 jabs due to the plasmid DNA contamination: https://www.youtube.com/watch?v=IEWHhrHiiTY
@joshg99 - Josh Guetzkow
And a transcript of his interview with Maryanne Demasi: https://maryannedemasi.substack.com/p/exclusive-an-interview-with-buckhaults

@joshg99 - Josh Guetzkow
Great talk by @hedleyrees discussing the intricacies and challenges of biologic drug manufacturing: https://rumble.com/v3fqujk-challenges-in-manufacturing-and-distributing-products-derived-from-biologic.html


@joshg99 - Josh Guetzkow
Remember how the COVID vaccines contaminated with endotoxin & plasmid DNA that Pfizer/BioNTech sold to the world were only tested on 252 people, but they said they'd compare them w/the ~20K subjects who got higher quality ones? New FOIA proves they never did that! But I did.🧵 https://t.co/ONgaR2OxgU
@joshg99 - Josh Guetzkow
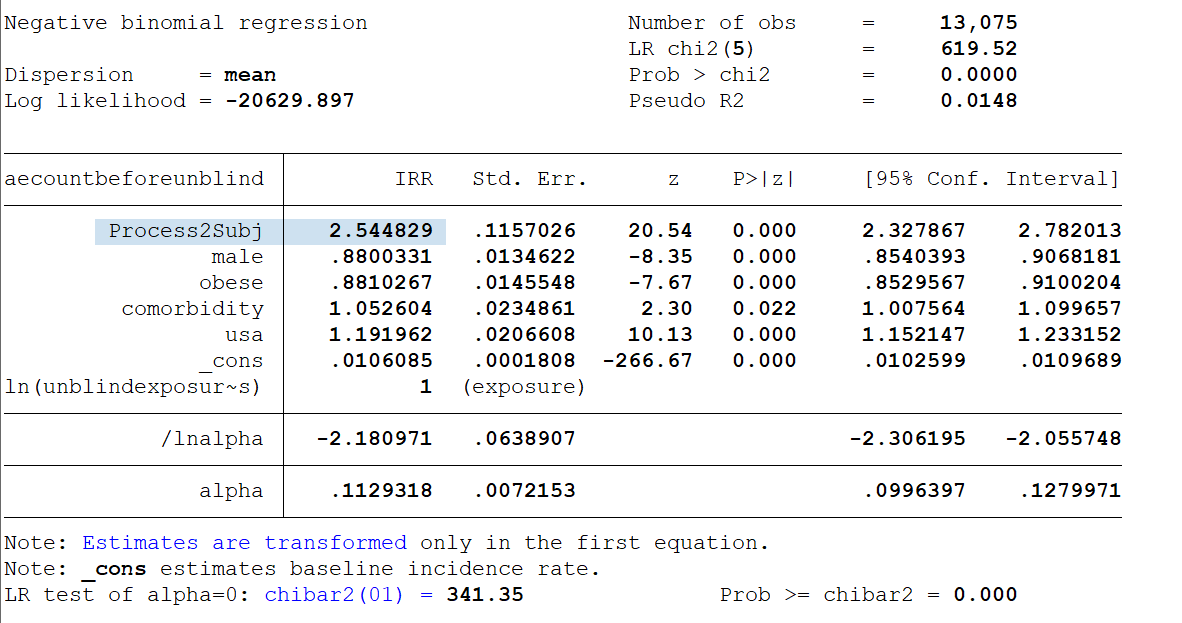
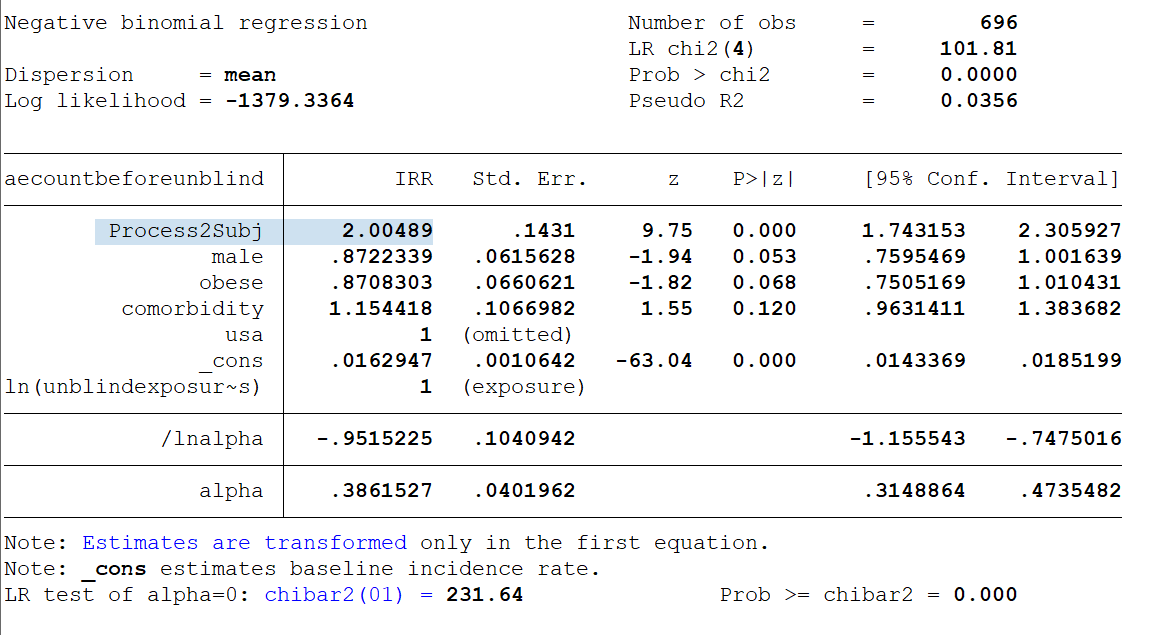
The adverse event rate among the subjects who got the new 'Process 2' doses was 2.5x higher than the AE rate of all other treatment subjects in the same age group & 2x higher than other treatment subjects at the same sites -- controlling for sex, obesity and comorbidities. https://t.co/EqKIoRSfq6
@joshg99 - Josh Guetzkow
Only 4 of the process 2 subjects were tested for neutralizing antibody titers (aka immunogenicity) -- all of them aged 22 and younger. Only 3 out of the 4 showed presence of antibody titers 1 month after second jab. None were tested for neutralizing titers before vaccination. https://t.co/aGqFjgjy4o
@joshg99 - Josh Guetzkow
What was the excuse Pfizer/BioNTech gave in Sep. 2022 for shirking their responsibility to do the comparison study by Feb 2021 as stated in their protocol? No need due to the "extensive usage of vaccines manufactured via 'Process 2'" as of Sept 2022. Make it make sense! https://t.co/uW3hKo7uMY
@joshg99 - Josh Guetzkow
For background and more information on the Pfizer/BioNTech bait-and-switch scandal, see this thread. 👇 https://x.com/joshg99/status/1658421192326365185
@joshg99 - Josh Guetzkow
The FOIA was obtained by Perseus Group member @NickHunt5, who also writes for the Daily Sceptic. Here is the cover page of the MHRA response to his request (FOI# 23/510): https://t.co/YgvJh2ZbFc
@joshg99 - Josh Guetzkow
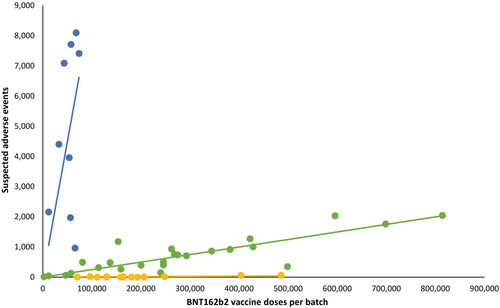
An easy way to remember the key difference between process 1 and 2: https://t.co/H8HCYZ54xb